Clinical & Medical Surgery
Open Access
Volume 5
Aierxiding Aimaiti, BS*
*Corresponding Author: Zhiqiang Zhao
Department of Musculoskeletal Oncology Center, The First Affiliated Hospital, Sun Yat-Sen University, 58 Zhongshan 2nd Rd, Guangzhou, Guangdong, China.
Email: zhaozhq7@mail.sysu.edu.cn
Article Info
Received: Oct 24, 2025
Accepted: Nov 21, 2025
Published: Nov 28, 2025
Archived: www.jclinmedsurgery.com
Copyright: © Aimaiti A (2025).
Abstract...
Purpose: This study aimed to validate potential clinical and radiological features for distinguishing enchondromas from Atypical Cartilaginous Tumours (ACT) and to analyse the association between maximum tumour diameter and local recurrence of enchondromas.
Materials and methods: This retrospective study reviewed the clinical data of 50 patients pathologically diagnosed with enchondroma or ACT and treated in our center between 1 January 2015 and 1 June 2024. Imaging characteristics, recurrence outcomes and other variables were compared among patients, and chi-square tests were used to assess the influence of maximum tumour diameter and other relevant factors on the recurrence rate.
Results: Patients with a maximum tumor diameter ≥5 cm had a significantly higher recurrence risk (recurrence 8% vs. non-recurrence 0%, P=0.01, P<0.05). This difference is not only statistically significant but also aids in distinguishing enchondroma from ACT. Moreover, all recurrent cases exhibited cortical bone destruction, further highlighting this imaging feature as a key differential point. The primary treatment consisted of curettage combined with bone grafting, yielding an overall favorable prognosis.
Discussion: A maximum tumor diameter of ≥5 cm and cortical bone destruction are not only significant predictors of local recurrence but also serve as key indicators in distinguishing Atypical Cartilaginous Tumors (ACTs) from enchondromas. Based on our findings, we are more inclined to consider lesions ≥5 cm as ACTs, particularly when accompanied by cortical destruction, as these features suggest a more aggressive biological behavior. Therefore, we recommend performing extended curettage combined with intraoperative electrocautery or other effective local adjuvant techniques in such cases to achieve adequate tumor control and reduce the risk of recurrence.
Keywords: Enchondroma; Atypical cartilaginous tumor; Maximum tumor diameter; Cortical destruction; Extended curettage; Local recurrence; Long bones.
Abbreviations: ACT: Atypical Cartilaginous Tumor; PMMA: Polymethyl-Methacrylate; MSTS: Musculoskeletal Tumor Society; MRI: Magnetic Resonance Imaging; CT: Computed Tomography.
Citation: Aimaiti A. Maximum tumor diameter ≥5 cm with cortical breach defines recurrence risk and distinguishes atypical cartilaginous tumors from enchondroma in long bones. J Clin Med Surgery. 2025; 5(2): 1218.
Introduction
Cartilaginous tumors of long bones encompass a spectrum of entities ranging from benign enchondromas to low-grade malignant Atypical Cartilaginous Tumors (ACTs). Despite their histopathological similarities, enchondromas and ACTs differ significantly in biological behavior and clinical management [1,2]. Enchondromas are typically indolent and often managed conservatively, whereas ACTs—considered grade 1 chondrosarcomas under the current WHO classification—exhibit locally aggressive behavior and require surgical intervention [3]. Consequently, accurate discrimination between the two is essential [4].
Accurate differentiation between these two entities is essential to avoid both undertreatment and overtreatment. However, this remains a diagnostic challenge due to overlapping histological features, such as mild nuclear atypia, occasional binucleation, and low mitotic activity [5-7]. Consequently, integration of clinical, radiological, and pathological data is necessary to establish a definitive diagnosis [1,8].
Previous studies have identified several imaging parameters that may aid in distinguishing ACTs from enchondromas, including tumor size, endosteal scalloping, cortical destruction, and the presence of soft tissue edema or extracompartmental extension [9-11]. Among these, tumor diameter has emerged as a potentially reliable predictor of biological aggressiveness. Gassert et al. proposed a threshold of 5 cm as a discriminatory cutoff between benign and malignant cartilaginous lesions in long bones [12]. However, the prognostic significance of this parameter, particularly in predicting local recurrence, remains underexplored.
In this retrospective study, we aimed to evaluate the role of maximum tumor diameter and cortical bone destruction in predicting local recurrence and facilitating the differential diagnosis between enchondromas and ACTs in long bones.
Methods
This was a single-center retrospective study. We consecutively enrolled 50 patients with postoperatively pathologically confirmed long-bone enchondroma or ACT who underwent surgery at our Musculoskeletal Oncology Center between January 2015 and June 2024. Individuals with concurrent malignant neoplasms or incomplete clinical/imaging data were excluded. The institutional ethics committee approved the protocol and waived informed consent.
Demographics (age, sex) were collected. Pre-operative radiographs, CT and MRI were used to measure maximum tumor diameter and to evaluate margin definition, cortical breach, periosteal reaction, etc. Patients were stratified into <5 cm and ≥5 cm groups using the 5 cm cutoff proposed by Gassert et al. [12]. Histopathologic specimens were re-reviewed by a musculoskeletal pathologist for cellularity, nuclear atypia and invasion.
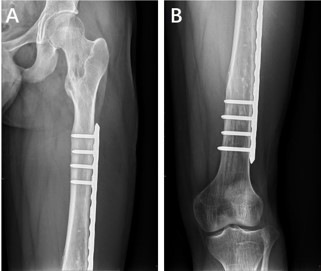
All lesions were treated with extended curettage: the cavity was enlarged 2-3 mm beyond the radiologic margin with a high-speed burr, the wall was cauterized with a high-frequency electrosurgical electrode and rinsed with saline, and the defect was reconstructed with autologous cancellous bone or PMMA cement. Prophylactic plate or intramedullary fixation was added when >50 % of the cortex was absent or in high-load regions.
Patients were followed radiographically at 3, 6 and 12 months post-operatively and every 6-12 months thereafter. Local recurrence was defined as reappearance of tumor on imaging confirmed by biopsy or secondary surgery. Statistical analysis was performed with SPSS 26.0; categorical variables were compared using Fisher’s exact test and continuous variables with the Mann–Whitney U test, with P<0.05 considered significant.
Results
Among the 50 long-bone cartilaginous lesions, 16(32%) had a maximum diameter ≥5 cm and 34 (68%) <5 cm; eight patients (16%) presented with multifocal disease, while 42(84%) had a solitary lesion. Solitary tumors were distributed as follows: femur 21 cases (50%), humerus 19 cases (45%), and tibia 2 cases
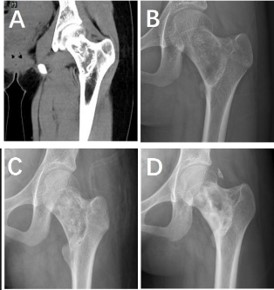
(5%). Age ranged from 1 to 66 years (mean 32.4 years). During postoperative follow-up, four patients (8%) developed local recurrence, all in the ≥5 cm group, and preoperative imaging in each showed cortical bone destruction. On MRI, recurrent lesions exhibited predominantly T2-hyperintense signal with sparse, punctate or patchy calcification (Table 1, Figure 1). Preoperative imaging revealed intramedullary, expansile cyst-like bone destruction located in the metaphysis or diaphysis of all lesions; 11 patients (22%) underwent prophylactic plate or intramedullary nailing because of cortical loss >50% or involvement of weight-bearing regions (Figure 2). Among the eight multifocal cases, one had synchronous lesions in the inferior right iliac wing, ischium, proximal and distal femur, and proximal tibia, and developed leg-length discrepancy after multiple surgeries. Postoperative pathological review showed slightly increased cell proliferation in five cases, of which three were ultimately diagnosed as ACT.
Table 1: Clinical characteristics of 50 patients.
| Characteristic | Study patients (N=50) | Recurrence | No recurrence | P-value |
|---|---|---|---|---|
| Gender | 0.621 | |||
| Male | 22(44%) | 1(2%) | 21(42%) | |
| Female | 28(48%) | 3(6%) | 25(50%) | |
| Tumor diameter | 0.010 | |||
| Less than 50 mm | 34(68%) | 0(0%) | 34(68%) | |
| Greater than or equal to 50 mm | 16(32%) | 4(8%) | 12(24%) | |
| Tumor location | 0.761 | |||
| Humerus | 18(36%) | 2(4%) | 16(32%) | |
| Femur | 29(58%) | 2(4%) | 27(54%) | |
| Tibia | 3(6%) | 0(0%) | 3(6%) | |
| Solitary/Multiple | 0.514 | |||
| Solitary | 42(84%) | 3(6%) | 39(78%) | |
| Multiple | 8(16%) | 1(2%) | 7(14%) | |
| Cortical bone destruction | 0.078 | |||
| Yes | 26(52%) | 4(8%) | 22(44%) | |
| No | 24(48%) | 0(0%) | 24(48%) | |
Discussion
Cartilaginous tumors of long bones form a continuous spectrum from benign enchondroma to low-grade Atypical Cartilaginous Tumor (ACT). Histologically, the two entities overlap considerably in cellularity, nuclear atypia and binucleation, so small biopsy samples often lead to underestimation or misdiagnosis [8,13,14]. The WHO 2020 classification defines ACT as a “locally aggressive, grade 1 chondrosarcoma without metastatic potential,” yet its diagnosis still requires integration of imaging, pathology and clinical findings, and no single gold standard exists [3,15].
In recent years, several retrospective studies have proposed that CT/MRI features such as diameter ≥5 cm, cortical destruction or scalloping >2/3, soft-tissue edema, calcification occupying <1/3 of the matrix, and a continuous growth pattern can help distinguish ACT [6,14,16]. Gassert et al. [14] validated these criteria in 82 long-bone lesions and found significantly increased odds ratios when they were combined, but sensitivity and specificity remain insufficient for routine clinical decisions. In our consecutive cohort of 50 long-bone lesions, we reproduced the strong association between “≥5 cm plus cortical destruction” and local recurrence (8% vs 0%, p=0.01), providing independent validation of this threshold. Moreover, recurrent lesions showed heterogeneous T2 signal and sparse, punctate calcification, correlating with histological upgrade (three of five finally diagnosed as ACT), further supporting a hierarchical imaging-pathology correspondence.
This study, conducted in a single-center Chinese long-bone series, confirms the predictive value of the 5 cm cutoff for recurrence risk; incorporates “cortical destruction” into a combined criterion; and proposes a simple “large-diameter plus cortical breach” binary model that can be applied intra-operatively. We also provide detailed technical notes on extended curettage coupled with electrocautery and PMMA reconstruction, which yielded a low recurrence rate.
Based on our findings, we advocate managing long-bone cartilaginous lesions ≥5 cm in maximum diameter with cortical destruction as ACT: perform extended curettage with high-speed burr and high-frequency electrocautery cavity ablation, add prophylactic plate or intramedullary nailing when cortical loss exceeds 50% or in weight-bearing regions, and utilize the thermal effect of PMMA cement to further reduce residual tumor burden. Postoperative imaging surveillance every 3-6 months is recommended; any recurrence or histological upgrade should prompt conversion to wide resection [17-19]. Conversely, typical enchondromas <5 cm without cortical destruction can be managed conservatively, with surgery reserved for lesions that become painful or enlarge >6 mm within 3 years [20].
Our study has inherent limitations: its retrospective, singlecenter design, the small number of recurrence events, and heterogeneous follow-up intervals precluded multivariate survival analysis. Future multicenter prospective registries that incorporate texture analysis, radiomics, and molecular markers such as IDH1/2 mutations are needed to refine discrimination accuracy and define optimal intervention timing.
Conclusion
In summary, we validated “≥5 cm plus cortical destruction” as a simple and effective combination for distinguishing ACT from enchondroma in long bones. Extended curettage with local adjuvant ablation achieved excellent local control while preserving function, offering new evidence for individualized clinical decision-making.
Declarations
Conflict of interest: The authors declare no conflicts of interest.
Funding sources: No external funding was received.
Acknowledgements: We thank the radiology and pathology departments for multidisciplinary case review.
References
- Welkerling H, et al. A reproducible and simple grading system for classical chondrosarcomas: Analysis of 35 chondrosarcomas and 16 enchondromas with emphasis on recurrence rate and radiological and clinical data. Virchows Arch. 2003; 443: 725–33.
- Ferrer-Santacreu EM, et al. Enchondroma versus chondrosarcoma in long bones of appendicular skeleton: Clinical and radiological criteria—a follow-up. J Oncol. 2016; 2016: 8262079.
- Ferrer-Santacreu EM, et al. Enchondroma versus low-grade chondrosarcoma in appendicular skeleton: Clinical and radiological criteria. J Oncol. 2012; 2012: 437958.
- Schwab JH, et al. Does local recurrence impact survival in lowgrade chondrosarcoma of the long bones? Clin Orthop Relat Res. 2007; 462: 175–80.
- van der Geest IC, et al. Oncological and functional results of cryosurgical therapy of enchondromas and chondrosarcomas grade 1. J Surg Oncol. 2008; 98: 421–6.
- Choi BB, et al. MR differentiation of low-grade chondrosarcoma from enchondroma. Clin Imaging. 2013; 37: 542–7.
- Sheth DS, et al. Chondrosarcoma of the pelvis: Prognostic factors for 67 patients treated with definitive surgery. Cancer. 1996; 78: 745–50.
- Murphey MD, et al. Enchondroma versus chondrosarcoma in the appendicular skeleton: Differentiating features. Radiographics. 1998; 18: 1213–37.
- Crim J, et al. Can imaging criteria distinguish enchondroma from grade 1 chondrosarcoma? Eur J Radiol. 2015; 84: 2222–30.
- Douis H, et al. What are the differentiating clinical and MRI features of enchondromas from low-grade chondrosarcomas? Eur Radiol. 2018; 28: 398–409.
- Lee S, Yoon MA. Assessment of central cartilaginous tumor of the appendicular bone: Inter-observer and intermodality agreement and comparison of diagnostic performance of CT and MRI. Acta Radiol. 2022; 63: 376–86.
- Geirnaerdt MJ, et al. Usefulness of radiography in differentiating enchondroma from central grade 1 chondrosarcoma. AJR Am J Roentgenol. 1997; 169: 1097–104.
- Eefting D, et al. Assessment of interobserver variability and histologic parameters to improve reliability in classification and grading of central cartilaginous tumors. Am J Surg Pathol. 2009; 33: 50–7.
- Laitinen MK, et al. The role of grade in local recurrence and the disease-specific survival in chondrosarcomas. Bone Joint J. 2018; 100-b: 662–6.
- Lalam R, et al. ESSR consensus document for detection, characterization, and referral pathway for tumors and tumorlike lesions of bone. Semin Musculoskelet Radiol. 2017; 21: 630–47.
- Lisson CS, et al. Diagnostic value of MRI-based 3D texture analysis for tissue characterization and discrimination of low-grade chondrosarcoma from enchondroma: A pilot study. Eur Radiol. 2018; 28: 468–77.
- Funovics PT, et al. Low-grade chondrosarcoma of bone: Experiences from the Vienna Bone and Soft Tissue Tumour Registry. Int Orthop. 2011; 35: 1049–56.
- Hickey M, et al. A systematic review and meta-analysis of intralesional versus wide resection for intramedullary grade I chondrosarcoma of the extremities. Ann Surg Oncol. 2011; 18: 1705–9.
- Chen X, et al. Is intralesional resection suitable for central grade 1 chondrosarcoma: A systematic review and updated metaanalysis. Eur J Surg Oncol. 2017; 43: 1718–26.
- Sampath Kumar V, et al. Surveillance of intramedullary cartilage tumours in long bones. Bone Joint J. 2016; 98-b: 1542–7.