Clinical & Medical Surgery
Open Access
Volume 3
Parvathi Devi GK1; Arundhathi S2*
*Corresponding Author: Arundhathi S
Associate Professor of Pathology, All India Institute of Medical Sciences, Mangalagiri, Andhra Pradesh, India.
Email: arundhathi19@yahoo.co.in
Article Info
Received: Apr 17, 2023
Accepted: May 11, 2023
Published: May 18, 2023
Archived: www.jclinmedsurgery.com
Copyright: © Arundhathi S (2023).
Citation: Devi GKP, Arundhathi S. Synchronous Malignant Gastrointestinal Stromal Tumor of Ileum and Invasive Ductal Carcinoma of Breast: A Rare Case Report. J Clin Med Surgery. 2023; 3(1): 1095.
Introduction
Two or more tumors appearing simultaneously or consecutively in different organs are known as synchronous tumors [1]. Gastrointestinal stormal tumors (GISTs) are rare mesenchymal neoplasms of GIT with an incidence of 1.5/100,000/year [2]. Synchronous occurrence of GIST and breast carcinoma accounted for 7% of all GIST associated cancers [3]. We report a case of malignant GIST of ileum associated with left breast carcinoma in a 56 year old lady, who presented with mass in left breast.
Case report
A 56 year old female was admitted with complains of left breast mass of two months duration. She gave history of hysterectomy 10 years back. She did not have any abdominal symptoms at presentation. On examination, a mass measuring 5 x 3 cms was noted in left upper and outer quadrant of left breast, with a few left axillary lymph nodes.
Hematological investigations revealed that her hemoglobin was 7.6 g/dl and she had neutrophilic leukocytosis. Serum CA125 was normal. Echocardiography showed a small interatrial septal aneurysm. CT scan findings showed a lobulated hypodense lesion in upper outer quadrant of left breast measuring 57 x 37 x 45 mm and a few axillary lymph nodes. CT scan of pelvis showed an ill defined heterogeneously enhancing soft tissue lesion between urinary bladder and rectum which measured 77 x 57 x 49 mm. The final impression on contrast CT was carcinoma left breast and left axillary lymphadenopathy with a mass lesion in between bladder and rectum? pedunculated fibroid/ ? metastatic deposit/ ? GIST.
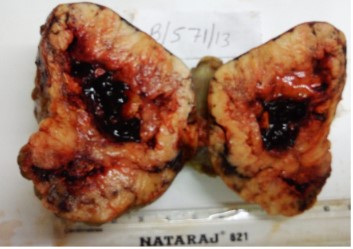
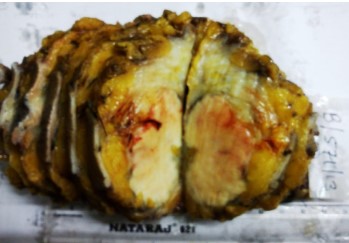
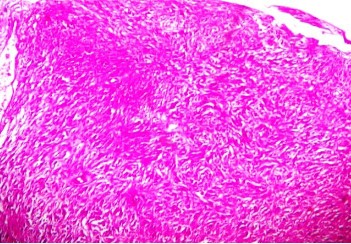
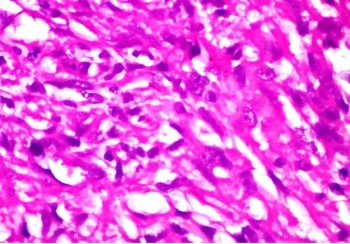
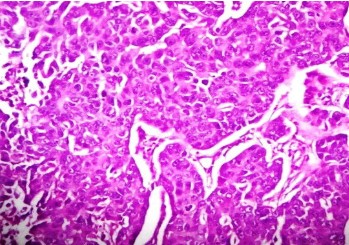
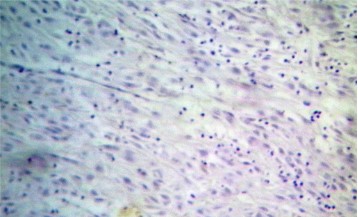
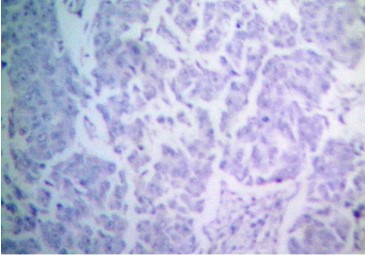
Fine needle aspiration cytology (FNAC) of left breast lump showed features of carcinoma. The final pre-operative diagnosis in conjunction with radiological and FNAC findings was carcinoma left breast stage III T3N1M0 with perforated proximal ileal mass? GIST. The patient was posted for surgery. The small intestinal perforated tumor was resected along with omentum, right and left ovary. She also underwent left radical mastectomy with axillary clearance. Post-operative period was uneventful. We received a small intestinal perforated tumor, a piece of omentum, right and left ovary. We found a lobulated mass measuring 8 x 5 x 4 cm which was attached to the wall of small intestine (ileum). The mass was pinkish white with areas of hemorrhage, necrosis and cavitary degeneration on cut section (Figure 1). We also received a piece of omentum measuring 4 x 3 x 0.5 cms, right ovary measuring 3 x 2 x 1 cms and left ovary measuring 2.5 x 2 x 1 cms. Cut surface of both ovaries were unremarkable. Also received left mastectomy specimen which on sectioning showed a grey white tumor measuring 5 x 4 x 3 cm in upper outer quadrant (Figure 2). We isolated 4 lymph nodes from the axillary pad of fat, largest measuring 1 x 1 x 1 cm, cut section of which were grey brown. Microscopically, small intestinal mass showed spindle cell tumor with spindle cells arranged in whorls and short fascicles (Figure 3). The cytoplasm is pale eosinophilic and the nuclei are ovoid with vesicular nuclei and prominent nucleoli in some cells (Figure 4). Abnormal mitotic figures were seen around 4-6/50hpf. Areas of hemorrhage were seen along with a few tumor giant cells (Figure 5). A few rounded cells with eosinophilic to clear cytoplasm and vesicular nucleus were also seen. Areas of dystrophic calcification were noted (Figure 6). Section from omentum showed deposits from the above tumor. Sections from right and left ovaries were unremarkable. Sections from left radical mastectomy specimen showed features of infiltrating ductal carcinoma with tumor cells in sheets, syncitial pattern and in ductular pattern separated by fibrous tissue stroma (Figure 9). Sections from axillary lymph nodes showed reactive hyperplasia. No deposits were seen in the lymph nodes.
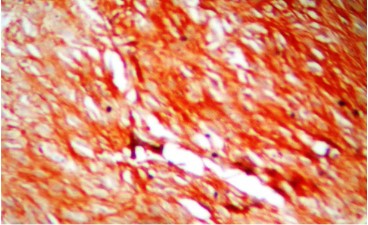
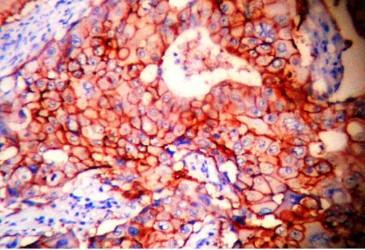
Immunohistochemistry (IHC) showed the tumor cells from the ileal mass were strongly positive for CD117/ c-KIT (Figure 7) and S-100 protein was negative (Figure 8), which confirmed it to be GIST. The carcinoma of breast was ER and PR negative but was positive for Her-2/neu which was 3+ in 65% of tumor cells (Figure 9). With this we concluded and confirmed that this was a case of synchronous occurrence of malignant ileal GIST with Her-2/neu positive IDC of left breast. The patient was referred to higher centre for forther treatment and follow up.
Discussion
Gastrointestinal stromal tumors (GISTs) are rare mesenchy- mal neoplasms of gastrointestinal tract accounting for 0.1-3% of all primary GI neoplasms and 5-10% of all sarcomas [4]. They originate from interstial cells of Cajal that regulate gut motility and this is proved by the expression of tyrosine kinase receptor KIT [5]. Majority of them existed in stomach (60-70%), followed by small intestine (20-25%), colorectum (5%) and esophagus (<5%) [6].
Benign GISTs outnumber malignant GISTs in the stomach, whereas malignant GISTs are more common in the intestines. Intestinal GISTs with maximum diameter of >5 cm or more than 5 mitoses/ 50 HPF are malignant [7].
GISTs (90- 95%) strongly express c- KIT (CD117) protein, 70% are positive for CD34, 30-40% for smooth muscle actin (SMA), 10% for S-100 and <5% for desmin [4].
The tumors that occur simultaneously or within 4 months-1 year of each other are known as synchronous tumors [1]. The literature search showed a few cases of synchronous tumors occurring concurrently with GISTs. The overall frequency of second tumors in different studies ranged between 4.5% and 33% [3]. A large study by Pandurengan et. al showed that 20% of patients with GIST developed other types of cancers [8]. The common GIST- associated cancers were gastrointestinal carcinomas (47%), followed by lymphoma/ leukemia (7%), carcinomas of prostate (9%), breast (7%), kidney (6%), lung (5%), female genital tract (5%) and carcinoid (3%) [3].
Regarding carcinogenesis of synchronous tumors of different histogenesis that coexist with GIST, various hypotheses have been postulated. Some authors have opined that they share common carcinogenic pathways or genetic mutations with proliferation of different cell lines [9]. Mutations in the KIT protooncogene are seen in 85- 90% of GISTs and 5-10% have mutations in PDGFRA. Mutations in KIT are also noted in chronic myeloid leukemia, germ cell tumors, small cell carcinoma of lung, neuroblastoma, melanoma, ovarian, breast and colorectal carcinoma [4]. Inspite of all these molecular studies, the occurrence of synchronous tumors as a mere coincidence cannot be ruled out.
Conclusion
The synchronous occurrence of GIST and breast carcinoma are well known, though infrequent. Further studies are essential to clarify the relationship of GIST with breast cancer and the role of c-KIT in the carcinogenesis of intraductal carcinoma of breast.
References
- Adim SB, Filiz G, Kanat O, Yerci O. Simultaneous occurrence of synchronous and metachronous tumors with gastrointestinal stromal tumors. Bratsil Lek Listy 2011; 112: 623-625.
- Nemes C, Rogojan L, Blaga TS, Seicean A, Dumitrascu DL, Ciuce C. Gastrointestinal stromal tumor (GIST) associated with synchronous colon adenocarcinoma- Acase report. J Gastrointestin Liver Dis. 2012; 21: 101-103.
- Agaimy A, Wunsch PH, Sobin LH, Lasota J, Miettiner M. Occurrence of other malignancies in patients with gastrointestinal stromal tumors. Semin Diagn Pathol. 2006; 23: 120-129.
- Arnogiannaki N, Martzoukou I, Kountourakis P, Dimitriadis E, Papathanasaki A, et. al. Synchronousbpresentation of GISTs and other primary neoplasms: A single centre experience. in vivo. 2010; 24: 109-116.
- Kindblom LG, Remotti HE, Aldenborg F and Meis- Kindblom JM. Gastrointestinal pacemaker cell tumor (GIPACT): Gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of Cajal. Am J Pathol. 1998; 152: 1259-1269.
- Miettiner M, Lasota J. Gastrointestinal stromal tumors- definition, clinical, histological, immunohistochemical and molecular genetic features and differential diagnosis. Virchows Arch. 2001; 438: 1- 12.
- Miettinen M, El. Rifai W, Sobin LH, Lasota J. Evaluation of malignancy and prognosis of gastrointestinal stromal tumours: a review. Human pathol. 2002; 33: 478- 483.
- Pandurengan RK, Dumont AG, Araujo DM, et.al. Survival of patients with multiple primary malignancies: a study of 783 patients with gastrointestinal stromal tumor. Ann Oncol 2010; 21: 2107-2111.
- Kaur R, Bhalla S, Nundy S, Jain S. Synchronous gastric gastrointestinal stromal tumor (GIST) and other primary neoplasms of gastrointestinal tract: report of two cases. Ann Gastroenterol. 2013; 26: 356-359.