Clinical & Medical Surgery
Open Access
Volume 3
Yi Guo1; Lei Yan2*; Huangjie Li1; Zhongcheng Wu1; Yuyin Duan3; Yanhai Meng4; Yuling An5
*Corresponding Author: Lei Yan
Department of Ultrasound, The First Affiliated Hospital of Fujian Medical University, 20# Chazhong Road, Fuzhou 350005, China.
Tel: 13705910071; Email: yanlei20082336@163.com
Article Info
Received: Apr 06, 2023
Accepted: May 02, 2023
Published: May 08, 2023
Archived: www.jclinmedsurgery.com
Copyright: © Yan L (2023).
Abstract...
Background: There is lack of outcomes of transaortic shallow septal myectomy with mitral valve(MV) repair comparing with extended Morrow procedure in treatment of hypertrophic obstructive cardiomyopathy (HOCM) with severe interstitial fibrosis.
Objectives: We report a 4-year single-center experience with transaortic shallow septal myectomy in combination with MV repair and compare it with extended Morrow surgery in a cohort of HOCM patients with severe interstitial fibrosis.
Methods: 36 patients who received surgery have been enrolled in current study. Their perioperative charac- teristics with echocardiographic results, myocardial histopathology and follow up outcomes had been graded and analyzed. We included two groups: 13 patients who received shallow septal myectomy concomitant with mitral valvuloplasty (MVP) due to the intrinsic abnormalities of MV apparatus (Shallow septal myectomy + MVP group), and 23 patients who only received extended Morrow procedure without any intrinsic MV abnormalities (Extended Morrow group).
Results: Preoperative results revealed that left ventricular end-diastolic dimension (LVEDD) (46.9 ± 1.41 mm vs. 11.4 ± 2.17 mm, p<0.05), posterior wall thickness (PWT) (13.3 ± 2.66 mm vs. 11.4 ± 2.17 mm, p<0.05), left ventricular mass (LVM) (440.2 ± 78.9 g Vs. 310.9 ± 127.6 g, p<0.05), left ventricular mass index (LVMI) (231.7 ± 75.39 g/m2 Vs. 180.2 ± 65.07 g/m2, p<0.05) and late gadolinium enhancement (LGE) (72.73% Vs. 27.27%, p<0.01) had showed the significant difference between the two group. In the myocardial histopathological evaluation, more severe interstitial fibrosis of the resected myocardium in Shallow septal myectomy + MVP group had showed statistical significant difference compared with Extended Morrow group (p<0.05). Shallow septal myectomy + MVP procedure sufficiently release left ventricular outflow tract obstruction(LVOTO) and decrease mitral regurgitation (MR) with no increase of postoperative arrhythmia compared with Extended Morrow surgery.
Citation: Guo Y, Yan L, Li H, Wu Z, Duan Y, et al. Transaortic Shallow Septal Myectomy with Mitral Valve Repair in Treatment of Hypertrophic Obstructive Cardiomyopathy with Severe Interstitial Fibrosis: Compared to Extended Morrow Procedure. J Clin Med Surgery. 2023; 3(1): 1092.
Introduction
Hypertrophic Obstructive Cardiomyopathy (HOCM) is a genetic disorder of the sarcomere characterized by asymmetric hypertrophy of the Left Ventricle (LV) [1]. Cardiomyocyte architecture is thought to be involved in arrhythmia in HOCM. Interstitial fibrosis was positively associated with ventricular arrhythmia and Atrial Fibrillation (AF) [1]. Septal myectomy in the extended Morrow procedure remains the gold standard and vital procedure for HOCM patients. The management of patients with severe interstitial fibrosis requires more attention. Late gadolinium enhancement (LGE) on MRI indicates severe interstitial fibrosis which have been closely associated with arrhythmia after septal myectomy [2].
On the other side, the surgical management of associated Mitral Valve (MV) lesions in HOCM resulting in Mitral Regurgitation (MR) is still a matter of debate. In HOCM, MV abnormalities can occur at any site of the MV [3]. Many abnormalities of the MV have been reported in the literature, in which the most common abnormalities are abnormally large/elongated mitral leaflets (especially Anterior Leaflets (AL)) and anterior displacement of any part of the mitral apparatus [4-14]. In fact, less pronounced septal basal hypertrophy may not be the only cause of left ventricular outflow tract obstruction (LVOTO), and this kind of clinical manifestation has been reported to be associated with leaflet and/or subvalvular apparatus abnormalities [15,16]. Regardless of Interventricular Septum (IVS) thickness, subvalvular apparatus remodeling combined with septal myectomy for patients with HOCM provides satisfactory long-term outcomes in terms of symptom improvement, LVOTO relief, and MR grade [17]. In this context, in order to eliminate the left ventricular outflow tract pressure gradient (LVOT PG) and restore MV function, it is advocated to correct MV abnormalities at the same time of septal myectomy [15,16,18-22]. In this study, compared with extended Morrow surgery, we sought to assess the clinical, echocardiographic and pathological results in patients requiring MV repair in addition to shallow septal myectomy.
Materials and methods
We retrospectively studied 36 consecutive cohorts of patients diagnosed with HOCM at Fuwai Yunnan Cardiovascular Hospital from September 2017 to September 2021, all of whomwere operated by a single surgeon and his team. This retrospective, single-center study was approved by the Ethics Committee of Fuwai Yunnan Cardiovascular Hospital (IRB2017-BG-028), and written informed consent was obtained from the patients. Indications for surgery include: (1) LVOT PG≥50 mmHg at rest or during physiological stimulation (mainly physiological exercise), as determined by Doppler transthoracic echocardiography (TTE); (2) The presence of drug-resistant symptoms. Medical treatment included beta-blockers and calcium channel blockers at the maximum tolerated dose, at least 6 months before surgery. At the same time, surgery was not recommended for patients who met the following criteria: (1) Patients older than 70 years of age, as our experience showed little improvement in symptoms and outcomes in these patients; (2) There is an additional risk of pump cardiac surgery, which may lead to early postoperative death.
We included 13 patients had Grade 1-3 MR who received transaortic shallow septal myectomy procedure concomitant with mitral valvuloplasty (MVP) due to the intrinsic abnormalities of the MV leaflet and/or subvalvular apparatus (shallow septal myectomy + MVP), compared with 23 patients had Grade 1-4 MR due to systolic anterior motion(SAM) who received extended Morrow procedure alone (Extended Morrow group).
Hospital records were reviewed using a dedicated database to analyze preoperative history and reports, surgical records, postoperative and follow-up echocardiography reports.
Preoperative imaging evaluation methods
All patients underwent preoperative TTE and MRI scan before surgery, both transesophageal echocardiography (TEE) during operation and gross valvular inspection during operation confirmed the function and severity of intrinsic abnormalities of MV.
The following basic variables were measured through pre-operative TTE: left ventricular outflow tract pressure gradient (LVOTPG), MR, left atrial dimension (LAD), left ventricular end-diastolic dimension (LVEDD), IVS thickness, posterior wall thickness (PWT), left ventricular mass (LVM), left ventricular mass index (LVMI), relative wall thickness (RWT), end-diastolic volume (EDV), end-diastolic volume index (EDVI), left ventricular ejection fraction (LVEF), cardiac output (CO), LVMI indication and RWT indication. The degree of MR was measured by TTE and TEE, and has been quantitatively graded as grade1 (mild), grade 2 (moderate), grade 3 (moderate-to-severe) and grade 4 (severe). Cardiac MRI provides detailed information on cardiac morphology, ventricular function, myocardial tissue characteristics, and LGE. Computed tomography (CT) was used to assess the presence of epicardial coronary artery disease (CAD) and/ or coronary artery bridges in patients with HOCM. Electrocardiogram was performed before and after operation to detect serious electrophysiological disorders such as heart block.
Surgical procedures
All procedures were performed with total sternotomy, aortic and right atrial cannulation, intermittent antegrade cold-blooded arrest with mild hypothermia (34OC).
In the Extended Morrow group, preoperative TTE, intraoperative TEE, or valvular examination by aortotomy was performed before septal myectomy, regardless of the extent of MR which was only associated with SAM. The resection range of septal myectomy in the Extended Morrow group was as follows: the upper end was located about 4 mm below the aortic ring, the lower end was extended to the apex of LV, the right end was started slightly rightward to the nadir of the right aortic cusp, and the left end was terminated near the mitral anterior commissure [23]. Myocardium excision need to be quite careful to ensure it go beyond the point of systolic mitral-septal contact [18,24].
In shallow septal myectomy + MVP group, about one-third of IVS thickness was removed by aortotomy. Bicaval cannulation and a left or right atrial approach was used in MVP. The ventricular surface of MV leaflet was systematically analyzed. Even if there were multiple anomalies of the MV in the HOCM, elongation of mitral leaflets is probably the most common anomaly. The fibrotic and retracted secondary chordae of the anterior MV leaflet were examined before resection. Fibrous or muscular structures that abnormally connect the papillary muscles (PM) to the septum or free wall were excised to increase the flexibility of PM. These procedures have the effect of reducing AL tether, thereby displacing the MV apparatus backward, away from the LVOT. Once elongated (slack) primary chordae had been observed after excision of the retracted secondary chordae from AL, which usually pertain to the central part of A2, the free edge of A2 needs to be folded laterally between the redundant chordae. If leaflet calcification or indentation is present, this should also be addressed so that the leaflet surface becomes smooth.
We then used TEE to confirm the decline of LVOT PG, SAM remission, and reduction in MR, followed by repeated direct pressure measurements after cardiopulmonary bypass (CPB) to assess outflow tract haemodynamics. CPB time, aorta interruption time and simultaneous operation have been analyzed as the operation data.
Histopathological methods
The length, width, height, and bulk of resected myocardium tissue was measured and analyzed in the examination of gross specimens from surgery. Meanwhile, specific hypertrophy localization was recorded and analyzed.
To assess histopathology, we extracted the extent of myocyte hypertrophy, myofibre disarray, interstitial fibrosis, and endocardial thickening from the histopathological report of the surgical myectomy specimen. Histological examination of resected myocardium was reviewed by a cardiovascular pathologist during routine clinical pathological examination. All slides were fixed with 10% neutral buffered formalin and stained with hematoxylin and eosin and treated with conventional clinical diagnostic protocols.
All cases were evaluated for the presence or absence of the following myocardial features: myocyte hypertrophy, myofiber disarray, interstitial fibrosis, and endocardial thickening. Hypertrophy can be diagnosed if the nucleus of myocyte is continuously enlarged and hyperchromatic. Using red blood cells as an internal indicator, the diameter of cardiomyocytes was approximately 3 red blood cells (RBC) for mild, 4 red blood cells for moderate, and 5 red blood cells for severe [1].
The myofiber disarray is characterized by cellular interlacing area, whirling area or herringbone pattern. If there is no any extent of the above three aspects then it is graded as none of myofiber disarray. If any of the above three aspects range from 1% to 25% of the myocardial area on the microscope slide, it is mild. Similarly, if it ranges from 26% to 50%, then it is moderate. If it is 50%, then it is severe [1].
We performed a semiquantitative, visual evaluation of interstitial fibrosis: none if there was absent of fibrosis, mild when the fibrosis is present less than 30% of the examined myocardium, moderate while the fibrosis is present between 30% to 60% of the examined myocardium, severe if the fibrosis is present more than 60% of the examined myocardium [25].
Endocardium thickness was evaluated for the presence of fibrotic thickening and regarded as none if the thickness was generally <30 μm, mild if 30 to 75 μm, moderate if 76 to 150 μm, and severe if >150 μm [1,26,27].
Follow-up
All patients underwent clinical and echocardiographic examination after operation. Postoperative TTE was performed in the first day after operation, prior to discharge, at 3 months, 6 months and 12 months in the first year, and then annually by same cardiologist from our echocardiography department. The following basic variables were measured through TTE: LAD, LVEDD, LVEF, IVS thickness in diastolic period, MR, LVOT PG and PWT. On March 31, 2022, 100% of patients were followed up, and the longest follow-up was 51 months.
Statistical analysis
When continuous variables fit a normal distribution, they were expressed as mean ± standard deviation and compared using an unpaired t-test. Conversely, if continuous variables did not fit a normal distribution, they were expressed as medians and interquartile ranges [50% (25-75%)] and compared with Mann Whitney test. Categorical variables are expressed as absolute numbers and/or percentages. All reported probability values are two-tailed p<0.05. SPSS 22.0 software was used for statistical analysis (SPSS Inc. Chicago, Ill).
Results
Preoperative outcome
Preoperative results between the two group are showed in Table 1. MR, LVEDD, PWT, LVM, LVMI and LGE in Shallow septal myectomy + MVP group had showed the significant difference between two group (p<0.05) compared with the Extended Morrow Group. The preoperative LVEF% and maximal IVS thickness showed no statistical differences between two groups (p<0.05). Figure 1 and 2 showed the details of the preoperative TTE and MRI image.
Perioperative outcome
In Shallow septal myectomy + MVP group of 13 patients, surgical myectomy was performed in all patients (100%). 12 patients (92.31%) were successfully underwent MVP, among whom one patient received concomitant dredging of right ventricular outflow tract (7.69%). One patient (7.69%) with the residual Grade 3 MR was performed mechanical mitral valve replacement (MVR) after 2 failed attempts to undergo MVP. MV remodeling by the main three procedures: (1) 8 patients (61.53%) received the edge-to-edge repair; (2) 7 patients (53.85%) received the resection of fibrotic, thickened, and agglutinated PM to the ventricular surface of AL; (3) 11 patients (84.62%) received resection of all anomalous trabeculae and accessory chordae tendineae. Due to complexity of MV abnormalities, there are 4 patients (30.77%) received only one procedure of MV repair; 7 patients (53.85%) were needed two procedures to repair; 2 patients (15.38%) received all three procedures. Nearly 70% cases have more than one MV procedure which make the surgery more complicated and need to be focused on MVP.
In extended Morrow group of 23 patients, five patients received the coronary artery bypass grafting (CABG) (21.73%), two patients received myocardial bridge release (8.70%), two patients received aortic valvuloplasty (8.70%), and TEE intraoperatively identified iatrogenic Ventricular Septal Defect (VSD) that was immediately repaired in one patient (4.35%).
Furthermore, CPB time was also increased in Shallow septal myectomy + MVP group due to MVP procedure (p<0.05). All the perioperative results showed in the Table 2. Intraoperative, in-hospital, and 30-day mortality were 0%.
Follow up results
The clinical follow up period was 6-51 months showed in Table 3, and significant reduction in peak resting LVOT PG in both two groups. During the follow up, maximal IVS thickness was significantly reduced in the both groups of surgical intervention, moreover thicker myocardium noted in Shallow septal myectomy + MVP group compared to Extended Morrow group (p<0.05). The LVEF and residual MR had showed the significant difference in the two groups (p<0.05). Third degree atrial ventricular block (AVB) with subsequent pacemaker implantation showed no statistical differences between two groups (p>0.05). One patient (7.69%) received permanent pacemaker implantation in Shallow septal myectomy + MVP group due to the severe injury resulting in bundle branch block. Two patients (8.70%) in Extended Morrow group received permanent pacemaker implantation before being discharged from the hospital: one patient was because of Complete Heart Block (CHB) on the basis of preoperative Complete Right Bundle Branch Block (CRBBB) and another patient has been progressed to postoperative CHB due to iatrogenic VSD during operation. There are no immediate postoperative atrial or ventricular arrhythmia, and also during the follow up period.
In Shallow septal myectomy + MVP group, there are 2 patients had new emerging mild aortic regurgitation (AR) during the whole follow-up, 4 patients had mild AR and 2 patients had mild -moderate AR in Extended Morrow group. Due to the small number of cases in which the new emerging AR occurred, no further statistical analysis of the above results was performed. All of the cases which developed AR due to the injury from aortotomy approach have been excluded from the study.
Intrinsic MV abnormalities in Shallow septal myectomy + MVP group
Identified through preoperative TTE and confirmed by intraoperative TEE or direct valve inspection, the MV apparatus anomalies generally were divided into 4 types: (1) annulus (n=2, 16.57%); (2) leaflets (n=8, 61.54%); (3) chordae tendineae (n=11, 84.62%) and (4) PM (n=4, 30.77%). The details of the sub-classification of every type of intrinsic abnormalities in MV was showed in Table 4.
Myocardium histopathological evaluation
The size of resected myocardium: The resected myocardium in length, width, height and bulk has no statistical differences between two groups as shown in Table 5. According to our study, there are no statistically significant difference in hypertrophy localization between the two groups.
Histopathological Evaluation
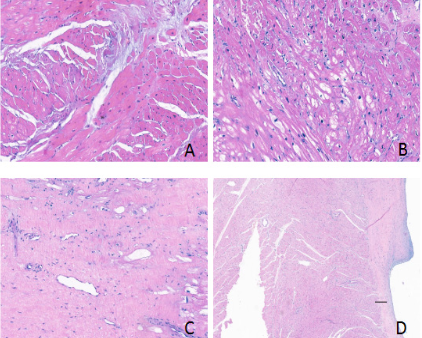
Most of the patients on both groups showed moderate or severe myocyte hypertrophy on histopathological examination. Other findings include myofibre disarray, interstitial fibrosis and endocardial thickening in both groups, which were categorized as none or mild degree. Interstitial fibrosis of the resected myocardium showed statistical significant in Shallow septal myectomy + MVP group Vs Extended Morrow group (p<0.05) in Table 5; moreover, this finding is consistent with LGE difference in preoperative MRI result as shown in Table 1. Figure 3 showed the histopathological imaging of myocyte hypertrophy, myofibre disarray, interstitial fibrosis and endocardial thickening in our study.
Table 1: Preoperative baseline clinical and demographic characteristics.
| Parameters | Shallow septal myectomy + MVP group | Extended morrow | P value |
|---|---|---|---|
| Preoperative | |||
| Age [years] | 49.9 ± 16.6 | 50.0 ± 14.3 | 0.9703 |
| Male | 69.2% (9/13) | 56.55% (13/23) | 0.4525 |
| NYHA functional class | 0.4365 | ||
| I[n] | 0 (0/13) | 30.4% (7/23) | |
| II[n] | 69.2% (9/13) | 34.8% (8/23) | |
| III[n] | 30.8% (4/13) | 34.8% (8/23) | |
| IV[n] | 0 (0/13) | 0 (0/13) | |
| Atrial fibrillation | 15.4% (2/13) | 8.70% (2/23) | 0.6092 |
| Echocardiography | |||
| LVOT PG [mmHg] | 61.7 ± 11.5 | 72.8 ± 25.8 | 0.1924 |
| MR | 0.0132 | ||
| Grade 1[n] | 2 | 13 | |
| Grade 2[n] | 5 | 7 | |
| Grade 3[n] | 6 | 2 | |
| Grade 4[n] | 0 | 1 | |
| LAD [mm] | 36.5 ± 4.98 | 34.9 ± 5.70 | 0.3693 |
| LVEDD [mm] | 46.9 ± 1.41 | 41.4 ± 3.53 | 0.0012 |
| Max IVS thickness [mm] | 25 (23-27) | 21 (18.5-28.5) | 0.1807 |
| PWT [mm] | 13 (12-14) | 11 (10-12) | 0.0182 |
| LVM [g] | 440.2 ± 78.9 | 310.9 ± 127.6 | 0.0012 |
| LVMI [g/m2] | 260.7 ± 4.6 | 180.2 ± 62.0 | 0.0015 |
| RWT [mm] | 0.5490 (0.5087-0.57855) | 0.5268(0.4558-0.6316) | 0.5858 |
| EDV [ml] | 111 (104.6-138.9) | 117(99.2-131.7) | 0.8262 |
| EDVI [ml/m2] | 72.6 ± 22.6 | 66.0 ± 16.3 | 0.3185 |
| LVEF [%] | 69.4 (66.6-75.3) | 74.2 (71.8-78.1) | 0.0587 |
| CO [L/min] | 6.0 ± 2.8 | 5.9±0.9 | 0.8678 |
| LVMI indication | 0.1363 | ||
| Normal range[n] | 0 | 2 | |
| Mildly enlarged[n] | 0 | 1 | |
| Moderate enlarged[n] | 0 | 2 | |
| Severely enlarged[n] | 18 | 18 | |
| RWT indication | >0.9999 | ||
| Concentric hypertrophy[n] | 13 | 23 | |
| Eccentric hypertrophy[n] | 0 | 0 | |
| LGE[%] | 72.73% | 27.27% | 0.0064 |
LVOTPG: Left Ventricular Outflow Tract Pressure Gradient; LAD: Left Atrial Dimension; LVEDD: Left Ventricular End-Diastolic Dimension; IVS: Interventricular Septum; PWT: Posterior Wall Thickness; LVM: Left Ventricular Mass; LVMI: Left Ventricular Mass Index; PWT: Posterior Wall Thickness; EDV: End-Diastolic Volume; EDVI: End-Diastolic Volume Index; LVEF: Left Ventricular Ejection Fraction; CO: Cardiac Output; LGE: Late Gadolinium Enhancement.
Table 2: Intraoperative characteristics.
| Parameters | Shallow septal myectomy + MVP group | Extended Morrow | P value |
|---|---|---|---|
| CPB [min] | 122.7 ± 41.3 | 94.7 ± 28.1 | 0.0212 |
| Aorta interruption time [min] | 73.8 ± 28.9 | 60.9 ± 18.8 | 0.1066 |
| Simultaneous operation | |||
| Dredging of right ventricular outflow obstruction [n] | 1 | 0 | |
| CABG [n] | 0 | 5 | |
| Myocardial bridge release [n] | 0 | 2 | |
| Aortic valvuloplasty | 0 | 2 | |
| Iatrogenic ventricular septal defect repair [n] | 0 | 1 | |
| Edge-edge repair [n] | 8 | 0 | |
| Papillary muscles remodeling [n] | 7 | 0 | |
| Chordae tendineae repair [n] | 11 | 0 |
CPB: Cardiopulmonary Bypass; CABG: Coronary Artery Bypass Grafting
Table 3: Postoperative and follow up characteristics.
| Parameters | Shallow septal myectomy + MVP group | Extended Morrow | P value |
|---|---|---|---|
| Preoperative | |||
| Age [years] | 49.9 ± 16.6 | 50.0 ± 14.3 | 0.9703 |
| Male | 69.2% (9/13) | 56.55% (13/23) | 0.4525 |
| NYHA functional class | 0.4365 | ||
| I [n] | 0 (0/13) | 30.4% (7/23) | |
| II [n] | 69.2% (9/13) | 34.8% (8/23) | |
| III [n] | 30.8% (4/13) | 34.8% (8/23) | |
| IV [n] | 0 (0/13) | 0 (0/13) | |
| Atrial fibrillation | 15.4% (2/13) | 8.70% (2/23) | 0.6092 |
| Echocardiography | |||
| LVOT PG [mmHg] | 61.7 ± 11.5 | 72.8 ± 25.8 | 0.1924 |
| MR | 0.0132 | ||
| Grade 1 [n] | 2 | 13 | |
| Grade 2 [n] | 5 | 7 | |
| Grade 3 [n] | 6 | 2 | |
| Grade 4 [n] | 0 | 1 | |
| LAD [mm] | 36.5 ± 4.98 | 34.9 ± 5.70 | 0.3693 |
| LVEDD [mm] | 46.9 ± 1.41 | 41.4 ± 3.53 | 0.0012 |
| Max IVS thickness [mm] | 25 (23-27) | 21 (18.5-28.5) | 0.1807 |
| PWT [mm] | 13 (12-14) | 11 (10-12) | 0.0182 |
| LVM [g] | 440.2 ± 78.9 | 310.9 ± 127.6 | 0.0012 |
| LVMI [g/m2] | 260.7 ± 4.6 | 180.2 ± 62.0 | 0.0015 |
| RWT [mm] | 0.5490 (0.5087-0.57855) | 0.5268(0.4558-0.6316) | 0.5858 |
| EDV [ml] | 111(104.6-138.9) | 117(99.2-131.7) | 0.8262 |
| EDVI [ml/m2] | 72.6 ± 22.6 | 66.0 ± 16.3 | 0.3185 |
| LVEF [%] | 69.4 (66.6-75.3) | 74.2 (71.8-78.1) | 0.0587 |
| CO [L/min] | 6.0 ± 2.8 | 5.9±0.9 | 0.8678 |
| LVMI indication | 0.1363 | ||
| Normal range [n] | 0 | 2 | |
| Mildly enlarged [n] | 0 | 1 | |
| Moderate enlarged [n] | 0 | 2 | |
| Severely enlarged [n] | 18 | 18 | |
| RWT indication | >0.9999 | ||
| Concentric hypertrophy [n] | 13 | 23 | |
| Eccentric hypertrophy [n] | 0 | 0 | |
| LGE [%] | 72.73% | 27.27% | 0.0064 |
LAD: Left Atrial Dimension; LVEDD: Left Ventricular End-Diastolic Dimension; LVEF: Left Ventricular Ejection Fraction; LVOT PG: Left Ventricular Outflow Tract Pressure Gradient; IVS: Intraventricular Septum; MR: Mitral Regurgitation; AR: Aortic Regurgitation; PWT: Posterior Wall Thickness
Table 4: Summarizes the intrinsic MV abnormalities in the shallow septal myectomy + MVP group.
| MV Apparatus | Abnormalities | n |
|---|---|---|
| Annulus | Typically large | 1(7.69%) |
| Annular calcification | 1(7.69%) | |
| Leaflets | Thickening and calcification in AL | 7(53.85%) |
| Thickening and calcification in PL | 4(30.77%) | |
| Prolapse | 3(23.08%) | |
| Clefts | 3(23.08%) | |
| Chordae tendineae | Elongated or fibrotic/retracted | 6(46.15%) |
| Potential for rupture of elongated chordae | 1(7.69%) | |
| Abnormal attachments to mitral leaflets | 3(23.08%) | |
| Attachment to the ventricular walls (false chords) | 0 | |
| PM | Hypertrophied | 3(23.08%) |
| Elongated or shortened | 0 | |
| Accessory PMs | 1(7.69%) | |
| Additional PM heads | 2(15.38%) | |
| PMs closer together, more anterior, medial, basal, or apical | 1(7.69%) | |
| Direct insertion to leaflet or fusion to the septum | 3(23.08%) |
Table 5: Baseline characteristics of the patients with myocardium histopathological evaluation.
| Shallow septal myectomy + MVP (n=13) | Extended Morrow (n=23) | P value | |
|---|---|---|---|
| Size of resected myocardium | |||
| Length [cm] | 4.00 ± 0.96 | 4.28 ± 1.10 | 0.4429 |
| Width [cm] | 2.96 ± 1.09 | 3.40 ± 0.94 | 0.209 |
| Height [cm] | 1.21 ± 0.77 | 1.03 ± 0.59 | 0.4426 |
| Bulk [cm3] | 16.69 (3.88-30) | 9.8 (7.35-27) | 0.7596 |
| Hypertrophy localization | >0.9999 | ||
| Septum [n] | 13 | 23 | |
| Lateral wall of LV [n] | 5 | 10 | |
| Posterior wall of LV [n] | 4 | 6 | |
| Anterior wall of LV [n] | 6 | 6 | |
| Apical of LV [n] | 1 | 2 | |
| Myocyte hypertrophy | 0.7839 | ||
| Mild [n] | 0 | 3 | |
| Moderate [n] | 6 | 5 | |
| Severe [n] | 7 | 15 | |
| Myocyte disarray | 0.204 | ||
| None [n] | 5 | 11 | |
| Mild [n] | 1 | 6 | |
| Moderate [n] | 3 | 4 | |
| Severe [n] | 4 | 2 | |
| Interstitial fibrosis | 0.0265 | ||
| None [n] | 3 | 12 | |
| Mild [n] | 3 | 7 | |
| Moderate [n] | 2 | 2 | |
| Severe [n] | 5 | 2 | |
| Endocardial thickening | 0.7644 | ||
| None [n] | 11 | 18 | |
| Mild [n] | 1 | 3 | |
| Moderate [n] | 1 | 2 | |
| Severe [n] | 0 | 0 | |
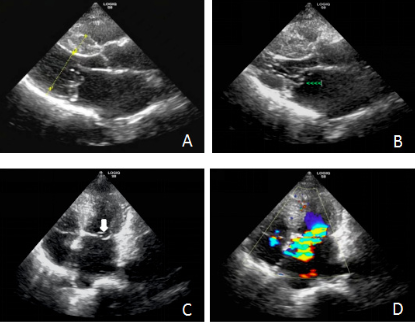
(A). Hypertrophic obstructive cardiomyopathy (HOCM) is showed in left ventricular long axis: interventricular septum(IVS) thickness is 1.84cm, anteroposterior diameter in left ventricular is 4.33 cm, and posterior wall thickness(PWT) in 0.72 cm
(B). Rupture of posterior chordae tendineae and mitral valve prolapse (green arrow) in left ventricular long axis
(C). Rupture of posterior chordae tendineae and mitral valve prolapse (white arrow) in four-chamber view
(D). Eccentric moderate mitral regurgitation (MR) (color).
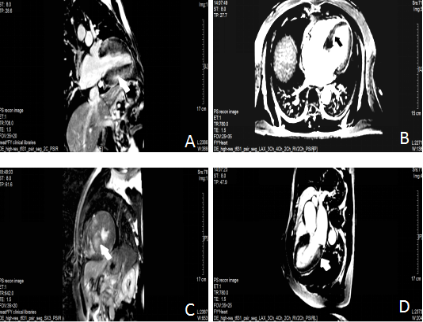
A. LGE is observed in the basal septal and inferior myocardium (white arrows).
B. LGE is observed in inferior myocardium (black arrows).
C. LGE is observed in the hypertrophied anterior septal myocardium (white arrows).
D. LGE is observed in the hypertrophied posterior myocardium (white arrows).
Discussion
The pathology and pathophysiology of HCM include hypertrophy of the LV with or without right ventricular hypertrophy, SAM, dynamic and mechanical LVOTO, MR, diastolic dysfunction, myocardial ischemia, and fibrosis [26]. Morrow and Brockenbrough first described septal myectomy as transaortic resection of partially hypertrophic basal IVS under CPB in 1961 [28]. Although septal resection alone was initially thought to be the sole etiology [29,30], it is clear recently that LVOTO is a dynamic process in which the changes of MV leaflet and/or the sub-val- vular apparatus play an important role [31-33]. These structural changes may directly determine the extent of LVOTO.
In daily practice, MV surgery as part of septal myectomy remains a matter of debate, depending on the morphology of the MV and the presence or absence of any intrinsic MV apparatus abnormalities. Adjuvant MV procedures include AL plication/ extension [31,34], reorientation of PM [35], secondary chordae cutting [18,36,37] and edge-to-edge repair [37,38], with the aim of completely relieving LVOTO by reducing the mobility of the AL and repositioning the MV apparatus posteriorly.
The current study compared 13 patients’ clinical data who underwent shallow septal myectomy and MVP with those of 23 patients who underwent extended Morrow procedure in the cardiac surgery department of Fuwai Yunnan Cardiovascular Hospital. Baseline clinical features and echocardiographic findings were similar to those of the China National Heart Center studies, with a mean age at 50 years of age and a predominance of men. We analyzed the perioperative clinical characteristics, myocardium histopathological features and follow up echocardiography findings of shallow septal myectomy with a concomitant MV procedure in our study, compared with the extended Morrow procedure. Preoperatively, maximal IVS thickness and LVOT PG have no significant statistical difference between the two groups in our study. However, we noticed that interstitial fibrosis of the resected myocardium is more severe in shallow septal myectomy + MVP group, which is in keeping with significant difference of preoperative LGE, LVEDD, PWT, LVM and LVMI between two groups in our study. All of this findings may be the indication of more severe LV remodeling in HOCM associated with intrinsic abnormalities in MV after long term LVOTO. Follow up results in our study suggest that shallow septal myectomy + MVP can effectively cut down the LVOT PG and decrease MR grade compared with extended Morrow procedure, with no increased risk of major arrhythmia (ventricular arrhythmia, emerging atrial fibrillation, conduction system injury with pace-maker implantation) after septal myectomy. Additional surgical repair of MV apparatus on top of shallow septal myectomy may reduce the need of a deep septal myectomy and also sufficiently release LVOTO with no increase in postoperative arrhythmia.
MR in HOCM is caused by poor coaptation of the posterior leaflet (PL) with the abnormally anteriorized AL, either due to insufficient length or restricted mobility, which results in an eccentric regurgitation jet to the posterior wall of the left atrium [39]. Our study showed that the main causes of abnormal tether of MV were thickening and calcification of the AL and elongated or fibrotic/retracted chordae tendineae. Meanwhile, there are half of cases have been reported to have more than one type of anomalies which make the surgery more complicated and need to focus on MVP, which has resulted in that 70% MVP has more than one procedure to repair the complex anatomy of MV intrinsic abnormalities.
Because of the intrinsic variability and complexity of HOCM, it is important to ensure that patients have received a thorough assessment and tailored intervention, including real-time echocardiographic assessment of LV geometry and MV- septum interaction. A better preoperative and intraoperative understanding of complex pathophysiology can translate into more predictable interventions and outcomes, which may help surgeons treat MV apparatus abnormalities [40].
In conclusion, shallow septal myectomy combined with MV repair has a good effect. This method has good clinical efficacy, especially for patients with severe interstitial fibrosis and left ventricular remodeling accompanied by LVOTO and intrinsic MV abnormalities.
Study limitations
This is a single center retrospective study. Further large-sample multicenter studies, as well as epidemiological studies, are needed to validate our findings and mechanisms. The statistical analysis was relatively simple. Limited by the short follow-up time and the small number of included cases, it is restricted to use Spearman rank correlation analysis to deeply explore in our study. Due to the limited number of patients enrolled in this study, neither ventricular arrhythmias nor emerging atrial fibrillation was confirmed, further studies are needed to reveal more details of arrhythmia between the two group.
Declarations
Ethical approval and consent: This study was in accordance with the Ethical Standards of the Institutional Ethics Committee of Fuwai Yunnan Cardiovascular Hospital and with the 1964 Helsinki declaration and its later amendments or comparable Ethical Standards. Written informed consent was provide by all included patients. Ethics batch number: IRB2017-BG-028.
Consent to publish: If this article is adopted, it is agreed to be published by all authors.
Funding information: 1. Yunnan Provincial Cardiovascular Disease Clinical Medical Center Project (No. FZX2019-06-01 2022YFKY008)
2. Fujian Provincial Science and Technology Planning Project social development science and technology guiding project (2022Y0010)
Conflict of interest: All authors declared that they have no conflicts of interest to this work. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
References
- Cui H, Schaff HV, Lentz Carvalho J, Nishimura RA, Geske JB, et al. Myocardial Histopathology in Patients With Obstructive Hypertrophic Cardiomyopathy. J Am Coll Cardiol. 2021; 77: 2159-2170.
- Olivotto I, Girolami F, Nistri S, Rossi A, Rega L, et al. The many faces of hypertrophic cardiomyopathy: from developmental biology to clinical practice. J Cardiovasc Transl Res. 2009; 2: 349-367.
- Jain CC, Newman DB, Geske JB. Mitral Valve Disease in Hypertrophic Cardiomyopathy:Evaluation and Management. Curr Cardiol Rep. 2019; 21: 136.
- Maron BJ, Maron MS, Wigle ED, Braunwald E. The 50-year history, controversy, and clinical implications of left ventricular outflow tract obstruction in hypertrophic cardiomyopathy from idiopathic hypertrophic subaortic stenosis to hypertrophic cardiomyopathy: from idiopathic hypertrophic subaortic stenosis to hypertrophic cardiomyopathy. J Am Coll Cardiol. 2009; 54: 191-200.
- Chaput M, Handschumacher MD, Tournoux F, Hua L, Guerrero JL, et al. Mitral leaflet adaptation to ventricular remodeling: occurrence and adequacy in patients with functional mitral regurgitation. Circulation. 2008; 118: 845-852.
- He S, Hopmeyer J, Lefebvre XP, Schwammenthal E, Yoganathan AP, et al. Importance of leaflet elongation in causing systolic anterior motion of the mitral valve. J Heart Valve Dis. 1997; 6: 149-159.
- Klues HG, Maron BJ, Dollar AL, Roberts WC. Diversity of structural mitral valve alterations in hypertrophic cardiomyopathy. Circulation. 1992; 85: 1651-1660.
- McIntosh CL, Maron BJ, Cannon RO, Klues HG. Initial results of combined anterior mitral leaflet plication and ventricular septal myotomy-myectomy for relief of left ventricular outflow tract obstruction in patients with hypertrophic cardiomyopathy. Circulation. 1992; 86: II60-7.
- Cooley DA. Surgical techniques for hypertrophic left ventricular obstructive myopathy including mitral valve plication. J Card Surg. 1991; 6: 29-33.
- Halpern DG, Swistel DG, Po JR, Joshi R, Winson G, et al. Echo-cardiography before and after resect-plicate-release surgical myectomy for obstructive hypertrophic cardiomyopathy. J Am Soc Echocardiogr. 2015; 28: 1318-1328.
- Jiang L, Levine RA, King ME, Weyman AE. An integrated mechanism for systolic anterior motion of the mitral valve in hypertrophic cardiomyopathy based on echocardiographic observations. Am Heart J. 1987; 113: 633-644.
- Maron MS, Olivotto I, Harrigan C, Appelbaum E, Gibson CM, et al. Mitral valve abnormalities identified by cardiovascular magnetic resonance represent a primary phenotypic expression of hypertrophic cardiomyopathy. Circulation. 2011; 124: 40-47.
- Sorajja P, Pedersen WA, Bae R, Lesser JR, Jay D, et al. First Experience With Percutaneous Mitral Valve Plication as Primary Therapy for Symptomatic Obstructive Hypertrophic Cardiomyopathy. J Am Coll Cardiol. 2016; 67: 2811-2818.
- Wan CK, Dearani JA, Sundt TM, Ommen SR, Schaff HV. What is the best surgical treatment for obstructive hypertrophic cardiomyopathy and degenerative mitral regurgitation? Ann Thorac Surg. 2009; 88: 727-731.
- Raffa GM, Romano G, Turrisi M, Morsolini M, Gentile G, et al. Pathoanatomic Findings and Treatment During Hypertrophic Obstructive Cardiomyopathy Surgery: The Role of Mitral Valve. Heart Lung Circ. 2019; 28: 477-485.
- Patel P, Dhillon A, Popovic ZB, Smedira NG, Rizzo J, et al. Left Ventricular Outflow Tract Obstruction in Hypertrophic Cardiomyopathy Patients Without Severe Septal Hypertrophy: Implications of Mitral Valve and Papillary Muscle Abnormalities Assessed Using Cardiac Magnetic Resonance and Echocardiography. Circ Cardiovasc Imaging. 2015; 8: e003132.
- Raffa GM, Franca E, Lachina C, Palmeri A, Kowalewski M, et al. Septal Thickness Does Not Impact Outcome After Hypertrophic Obstructive Cardiomyopathy Surgery (Septal Myectomy and Subvalvular Mitral Apparatus Remodeling): A 15-Years of Experience. Front Cardiovasc Med. 2022; 9: 853582.
- Lapenna E, Nisi T, Carino D, Bargagna M, Ruggeri S, et al. Hypertrophic cardiomyopathy with moderate septal thickness and mitral regurgitation: long-term surgical results. Eur J Cardiothorac Surg. 2021; 60: 244-251.
- Ferrazzi P, Spirito P, Iacovoni A, Calabrese A, Migliorati K, et al. Transaortic Chordal Cutting: Mitral Valve Repair for Obstructive Hypertrophic Cardiomyopathy With Mild Septal Hypertrophy. J Am Coll Cardiol. 2015; 66: 1687-1696.
- Sherrid MV, Balaram S, Kim B, Axel L, Swistel DG. The Mitral Valve in Obstructive Hypertrophic Cardiomyopathy: A Test in Context. J Am Coll Cardiol. 2016; 67: 1846-1858.
- Silbiger JJ. Abnormalities of the Mitral Apparatus in Hypertrophic Cardiomyopathy: Echocardiographic, Pathophysiologic, and Surgical Insights. J Am Soc Echocardiogr. 2016; 29: 622-639.
- Raffa GM, Pilato M. Hypertrophic Obstructive Cardiomyopathy and Subvalvular Mitral Apparatus Remodeling. Ann Thorac Surg. 2019; 108: 964.
- Wang S, Cui H, Yu Q, Chen H, Zhu C, et al. Excision of anomalous muscle bundles as an important addition to extended septal myectomy for treatment of left ventricular outflow tract obstruction. J Thorac Cardiovasc Surg. 2016; 152: 461-468.
- Iacovoni A, Spirito P, Simon C, Iascone M, Di Dedda G, et al. A contemporary European experience with surgical septal myectomy in hypertrophic cardiomyopathy. Eur Heart J. 2012; 33: 2080-2087.
- Galati G, Leone O, Pasquale F, Olivotto I, Biagini E, et al. Histological and Histometric Characterization of Myocardial Fibrosis in End-Stage Hypertrophic Cardiomyopathy: A Clinical-Pathological Study of 30 Explanted Hearts. Circ Heart Fail. 2016; 9: e003090.
- Raman B, Ariga R, Spartera M, Sivalokanathan S, Chan K, et al. Progression of myocardial fibrosis in hypertrophic cardiomyopathy: mechanisms and clinical implications. Eur Heart J Cardiovasc Imaging. 2019; 20:157-167.
- Varma PK, Neema PK. Hypertrophic cardiomyopathy: part 1 - introduction, pathology and pathophysiology. Ann Card Anaesth. 2014; 17: 118-124.
- MORROW AG, BROCKENBROUGH EC. Surgical treatment of idiopathic hypertrophic subaortic stenosis: technic and hemodynamic results of subaortic ventriculomyotomy. Ann Surg. 1961; 154: 181-189.
- BRAUNWALD E, LAMBREW CT, ROCKOFF SD, ROSS J, MORROW AG. IDIOPATHIC HYPERTROPHIC SUBAORTIC STENOSIS. I. A DESCRIPTION OF THE DISEASE BASED UPON AN ANALYSIS OF 64 PATIENTS. Circulation. 1964; 30: 3-119.
- BROCK R. Functional obstruction of the left ventricle; acquired aortic subvalvar stenosis. Guys Hosp Rep. 1957; 106: 221-238.
- Balaram SK, Ross RE, Sherrid MV, Schwartz GS, Hillel Z, et al. Role of mitral valve plication in the surgical management of hypertrophic cardiomyopathy. Ann Thorac Surg. 2012; 94: 1990-1997.
- Klues HG, Roberts WC, Maron BJ. Anomalous insertion of papillary muscle directly into anterior mitral leaflet in hypertrophic cardiomyopathy. Significance in producing left ventricular outflow obstruction. Circulation. 1991; 84: 1188-1197.
- Klues HG, Roberts WC, Maron BJ. Morphological determinants of echocardiographic patterns of mitral valve systolic anterior motion in obstructive hypertrophic cardiomyopathy. Circulation. 1993; 87: 1570-1579.
- Vriesendorp PA, Schinkel AF, Soliman OI, Kofflard MJ, de Jong PL, et al. Long-term benefit of myectomy and anterior mitral leaflet extension in obstructive hypertrophic cardiomyopathy. Am J Cardiol. 2015; 115: 670-675.
- Kwon DH, Smedira NG, Thamilarasan M, Lytle BW, Lever H, et al. Characteristics and surgical outcomes of symptomatic patients with hypertrophic cardiomyopathy with abnormal papillary muscle morphology undergoing papillary muscle reorientation. J Thorac Cardiovasc Surg. 2010; 140: 317-324.
- Bogachev-Prokophiev A, Afanasyev A, Zheleznev S, Fomenko M, Sharifulin R, et al. Mitral valve repair or replacement in hypertrophic obstructive cardiomyopathy: a prospective randomized study. Interact Cardiovasc Thorac Surg. 2017; 25: 356-362.
- Afanasyev AV, Bogachev-Prokophiev AV, Zheleznev SI, Sharifulin RM, Zalesov AS, et al. Edge-to-Edge Repair Versus Secondary Cord Cutting During Septal Myectomy in Patients With Hypertrophic Obstructive Cardiomyopathy: A Pilot Randomised Study. Heart Lung Circ. 2021; 30: 438-445.
- Lapenna E, Nisi T, Ruggeri S, Trumello C, Del Forno B, et al. Edge-to-Edge Mitral Repair Associated With Septal Myectomy in Hypertrophic Obstructive Cardiomyopathy. Ann Thorac Surg. 2020; 110: 783-789.
- El-Hamamsy I, Lekadir K, Olivotto I, El Guindy A, Merrifield R, et al. Pattern and degree of left ventricular remodeling following a tailored surgical approach for hypertrophic obstructive cardiomyopathy. Glob Cardiol Sci Pract. 2012; 2012: 9.
- Dorobantu LF, Iosifescu TA, Ticulescu R, Greavu M, Alexandrescu M, et al. Transaortic Shallow Septal Myectomy and Cutting of Secondary Fibrotic Mitral Valve Chordae-A 5-Year Single-Center Experience in the Treatment of Hypertrophic Obstructive Cardiomyopathy. J Clin Med. 2022; 11: 3083.