Clinical & Medical Surgery
Open Access
Volume 3
Elena Olearo1*; Giulio Fraternali Orcioni2; Luigi Garaventa3; Stefano Uccella3; Andrea Puppo1
*Corresponding Author: Elena Olearo
Department of Obstetrics and Gynecology, Azienda Ospedaliera Santa Croce e Carle, Cuneo, Italy.
Tel: 0039171642535; Email: eolearo@gmail.com
Article Info
Received: Jan 21, 2023
Accepted: Feb 10, 2023
Published: Feb 20, 2023
Archived: www.jclinmedsurgery.com
Copyright: © Olearo E (2023)
Abstract...
This is the case of an elderly woman who presented clinically aggressive recurrence of adenosarcoma without clear unfavorable prognostic factors at first diagnosis. Clinical treatment of patient including surgery and the process of histopathologic diagnosis is described. This case highlights that many aspects of biology of mixed epithelial-mesenchymal malignant uterine tumors needs to be better clarified and even if uterine adenosarcoma has been defined as low grade sarcoma, it can behave as a very aggressive and pleomorphic disease.
Citation: Solearo E, Orcioni GF, Garaventa L, Uccella S, Puppo A. Uterine Adenosarcoma: An Unpredictable Enemy. J Clin Med Surgery. 2023; 3(1): 1075.
Introduction
Uterine Adenosarcoma (UA) is a mixed tumor of the uterus consisting of a benign glandular epithelium and a malignant mesenchymal component, which was originally reported as Mullerian adenosarcoma by Clement and Scully [1]. UA represents 8% of all uterine sarcomas and 0,5% of uterine malignant tumors [2]. There is a misconception that this is an indolent, low-grade sarcoma, but at least 50% of patients will develop disease recurrence, particularly among patient with risk factors such as sarcomatous overgrowth [3], deep myometrial and lymphovascular space invasion. The rarity of the tumor and variety of clinical presentation can lead to misdiagnosis [4], underestimating the risk of recurrence, also in previously defined “low risk” cases.
We report the case of an elderly woman who presented clinically aggressive recurrence of adenosarcoma without clear signs of sarcomatous overgrowth or clear unfavorable prognostic factors at first diagnosis.
Case presentation
A 85-year-old woman was referred to our hospital on August 2022 due to weight loss in the last 3 months and discovery of multiple abdominal solid masses. She had a history of B-CLL, Paget disease, hypertension and hypercholesterolemia.
On January 2021 she underwent laparotomic hysterectomy and bilateral salpingo-oophorectomy in a different hospital due to metrorrhagia. The clinical finding was a necrotic protruding polyp from the uterine cervix and the final pathology report described Adenofibroma.
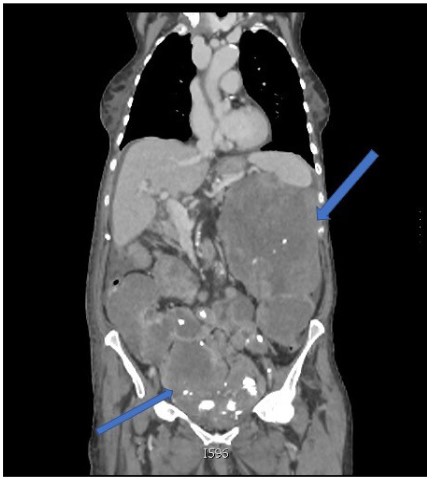
After surgery, she had no further assessment until she was readmitted in July 2022 for abdominal pain and distension. During hospital stay, increased CA 125 was documented (143,00 U/ml) and the CT-scan showed multiple large peritoneal solid masses, with calcific spots, involving upper abdomen and pelvis, with no signs of bowel obstruction, small amount of ascites and no signs of metastasis to abdominal organs or lungs (Figure 1). A CT guided biopsy of one of the peritoneal masses only showed mesenchymal cells and was inconclusive. She was then referred to our Hospital suspecting carcinosis from advanced ovarian cancer or sarcoma.
After reassessment, we performed mini-laparotomy with drainage of ascites and incisional biopsy of one of the masses. The pathological examination showed malignant spindle cell proliferation (CD10+, actin smooth muscle +, estrogen receptors 30%, beta-catenin -, progesterone receptor -, CKAE1/AE3 -, CD34 -, MDM-2 -, desmin -) with focal cytokaryological atypia and scattered mitotic figures. No epithelial components were found and ascites was negative for neoplastic cells.
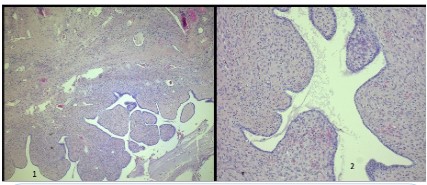
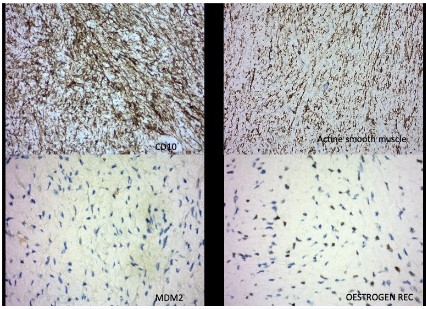
Since there were no clinical or pathological signs of non-gynecological mesenchymal tumors, the reassessment of the original uterine specimen from 2021 became mandatory. Revised pathological analysis of the uterine specimen revealed biphasic neoplasia with a polypoid profile consisting of areas with hyaline stroma circumscribed by benign cylindrical epithelium without cytokaryological atypia adenofibromatous-like, incorporating scattered hypercellulated areas with foci of necrosis, focal nuclear atypia of mild to moderate degree and scattered rare mitotic figures (Figure 2). Immunohistochemistry was performed for estrogen receptor (30%), progesterone receptor (negative), CD10, actina, MDM-2, beta catenina, CKAE1/AE3, CD34, S100 and desmina. CD10, actina and MDM-2 were positive in the stromal component (Figure 3).
The joint view of the two materials, i.e. the uterine lesion (2021) and the sample from solid masses (2022), allowed us to document that it was the same tumor; moreover, while a diagnosis of histological malignancy was intuitive on the specimen from the recurrence, a probative histological detail of malignant biological behavior on the uterine mass was not observed and it could be documented exclusively by the clinical behavior of the neoplastic elements arising from the stromal component.
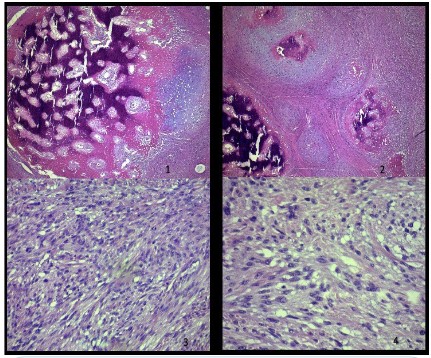
Therefore, the comparison of the specimens highlighted the presence of a uterine adenosarcoma, and samples from the recurrence showed “high grade” morphological aspects with small foci of heterologous stromal elements (osteochondrosarcoma Figure 4) consistent with the clinical evolution.
After diagnostic procedure, patient’s condition quickly worsened and she was readmitted for dyspnea and decay of general conditions due to the compressive effect of the masses. A new CT scan, showed pulmonary thromboembolism of the right pulmonary artery and overgrowth of the abdominal masses with the largest up to 26 cm, compressing iliac vessels. After informed consent with patient and care givers, we decided for palliative decompressive debulking surgery. Longitudinal xifopubic laparotomy revealed multiple sarcomatous masses strongly adherent to small bowel wall and sigmoid with infiltrative attitude. Carcinosis massively involved small bowel mesenter, mesorectum and peritoneum, liver and spleen. Given the palliative intent of the surgery, only the larger masses were removed, with massive adhesiolysis and multiple sutures on bowel wall. Postoperative course was complicated by delirium and small bowel perforation on day III after surgery, without acute abdomen or sepsis.
According to the poor prognosis given by the extent of the disease, patient general conditions and age, after large counselling with the patient and her family no further surgical or invasive treatment was undertaken and the patient was referred to palliative care.
Discussion
The WHO describes uterine adenosarcoma as a mixed epithelial and mesenchymal tumor in which the stromal component has a low malignant potential and the epithelial part is usually benign [5]. It more commonly appears after the menopause but there are several reports of UA in younger women of child bearing age. It is frequently described as a large protruding cervical polyp, thus leading to misdiagnosis [6,7]. As for uterine sarcomas, ultrasound imaging has no specific signs, therefore diagnosis rely on pathological evaluation. Macroscopically, the UA is a polypoid or multipolypoid lesion which can protrude through the endocervix and occupy the entire uterine cavity [5,8]. Microscopically, UA is characterized by benign glandular component and malignant stromal component [1]. The epithelial component has normally a benign endometrioid morphology though occasionally can have cellular atypia. The mesenchymal component is usually low grade [2]. When the sarcomatous component of the tumor reaches 25% of the total volume, it can be defined as sarcomatous overgrowth and it is linked to a worse prognosis [1-3]. Ten to 25% of adenosarcomas present heterologous stromal elements such as rhabdomyoblasts, liposarcoma, chondrosarcoma or smooth muscle-derived elements which increase the risk of recurrence and worsen prognosis [1,2,9]. Recurrences usually present only mesenchymal components at final pathology examination. Reported clinical risk factors for UA are endometriosis, history of pelvic irradiation, hyperestrogenism, use of tamoxifen, obesity and diabetes mellitus [2,10]. Risk of recurrence is about 50% in 10 years and it is increased by OS, myometrial invasion, lymphovascular invasion, cell necrosis and atypia, low mitotic count, and the presence of heterologous elements [2]. Surgical treatment (hysterectomy and bilateral salpingo-oophorectomy) represents the standard treatment for UA and it should be performed by an expert gynecologist oncologist in referral centres. Due to the low risk of regional lymph nodes metastasis, lymphadenectomy is not recommended [11] Currently there is no standardized specific radiation therapy, hormonal therapy or adjuvant chemotherapy for the treatment of UA. Adjuvant radiotherapy can be offered to low risk patient without sarcomatous overgrowth and less of 50% myometrial invasion [2-12]. Immunohistochemical markers commonly expressed by adenosarcoma are CD10 (71 to 100%) and WT1 (79%) while additional markers are smooth muscle actin (50 to 68%), CD34 (35%), desmin (32 to 62.5%), AE2/3 cytokeratin (25 to 27%) and vimentin (86%). EMA, Estrogen Receptor (ER), Progesterone Receptor (PR) are commonly expressed by the epithelial component [2]. Sarcomatous overgrowth is associated with the loss of expression of CD10, ER and PR [13]. SMARCA4-deficient uterine sarcoma has been recently described as a very aggressive entity that closely mimics the large cell variant of the small cell carcinoma of the ovary morphologically [14].
In summary, our patient did not show any significant clinical risk factors and we also investigated possible relationship between UA and B-CLL but none was found in literature [15]. The mixed tumor diagnosed on the uterine specimen did not show any significant risk factor or prognostic element to suspect such a malignant clinical behavior, neither immunohistochemistry was decisive since CD10 was still expressed, ER was 30% positive and only PR expression was lost, a condition inconsistent with those reported in literature for sarcomatous overgrowth.
Conclusion
Even if UA has been defined as low grade sarcoma, it can behave as a very aggressive and pleomorphic disease, therefore the main effort should be taken to have proper pathological diagnosis: due to its similarity to benign polyp, the missed diagnosis or the misdiagnosis is common in early stage of the disease [16]. Our case well represents how a mixed epithelial-mesenchymal malignant uterine neoplasm should not be considered a neoplasm with certainly biologically and clinically be- nign behavior, which is also documented by the deletion of the adenofibroma by the WHO classification [5].
We believe that this case may offer a chance to consider that any malignant mesenchymal lesion of the female pelvis could represents a malignant stromal component of a gynecologic biphasic neoplasm.
Finally, our case confirms that surgical management of early stage uterine adenosarcomas should not be modified according to the presence or absence of a “low” or "high grade" mesenchymal component and sarcomatous overgrowth, since it is not possible to stratify low or high-risk cases only according to molecular and cytogenetic data. Even the real impact of clinical, macro and microscopic characteristics (such as myometrial invasion, lymphovascular invasion, cellular necrosis and atypia, low mitotic count, and the presence of heterologous elements) will benefit from further research. Because of the rarity of the disease and lack of abundant case study reports, uniform clinical guidelines for treatment following surgical resection of a high-grade adenosarcoma remain unclear.
According to our experience, we would recommend to consider every patient with UA “at risk” and offer close follow-up or tailor adjuvant treatment when clinically indicated. Moreover, surgical and medical treatment at recurrence should always take into account patient age, clinical conditions and prognosis and be specifically tailored for each patient.
As rare tumor entity, we support the creation of national and international databases and network of specialists to exchange experiences and offer new perspectives to improve outcomes for UA.
Declarations
Funding: This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Written informed consent was obtained from the patient for the publication of this case report and accompanying images.
Declaration of competing interest: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgment: We thank Dr Rodolfo Brizio from Pathlogy Unit of ASL1 Imperia Hospital.
References
- Clement PB, Scully RE. Mullerian adenosarcoma of the uterus: a clinicopathologic analysis of 100 cases with a review of the literature. Hum Pathol. 1990; 21: 363-381.
- Nathenson MJ, Ravi V, Fleming N, Wang WL, Conley A. Uterine adenosarcoma: a review. Curr Oncol Rep. 2016; 18: 68.
- Krivak TC, Seidman JD, McBroom JW, MacKoul PJ, Aye LM, et al. Uterine adenosarcoma with sarcomatous overgrowth versus uterine carcinosarcoma: comparison of treatment and survival. Gynecol Oncol. 2001; 83: 89-94.
- Wang B, Yang HD, Shi XH, Li H. Advanced uterine adenosarcoma with sarcomatous overgrowth in a young woman: A case report. Medicine. 2019; 98: 47.
- WHO Classification of Tumours Editorial Board. Female Genital Tumours. Lyon: International Agency for Research on Cancer 2020.
- Verschraegen CF, Vasuratna A, Edwards C, Freedman R, Kudelka AP, et al. Clinicopathologic analysis of Mullerian adenosarcoma: the M.D. Anderson Cancer Center Experience. Oncol Rep. 1998; 5: 939-944.
- Pinto A, Howitt B. Uterine adenosarcoma. Arch Pathol Lab Med. 2016; 140: 286-290.
- McCluggage WG. A practical approach to the diagnosis of mixed epithelial and mesenchymal tumours of the uterus. Mod Pathol. 2016; 29: S78-91.
- Kaku T, Silverberg SG, Major FJ, Miller A, Fetter B, Brady MF. Adenosarcoma of the uterus: a Gynecologic Oncology Groupclinicopathologic study of 31 cases. Int J Gynecol Pathol. 1992; 11: 75-88
- Wang JH, Zhao J, Lin J. Opportunities and risk factors for premalignant and malignant transformation of endometrial polyps: management strategies. Minim. Invasive Gynecol. 2010; 17: 53- 58.
- Hu H, Wei Z, Zhao H, Yuan G. Epidemiology of adenosarcoma and the inverse probability of treatment weighting (IPTW) adjusted survival analysis of lymph node dissection in uterine adenosarcoma. Medicine. 2022; 23: 38
- Reed NS, Mangioni C, Malmström H, Scarfone G, Poveda A. Phase III randomised study to evaluate the role of adjuvant pelvic radiotherapy in the treatment of uterine sarcomas stages I and II: a European Organisation for Research and Treatment of Cancer Gynaecological Cancer Group Study (protocol 55874). Eur J Cancer 2008; 44: 808-818.
- Soslow RA, Ali A, Oliva E. Mullerian adenosarcomas: an immunophenotypic analysis of 35 cases. Am J Surg Pathol. 2008; 32: 1013-1021.
- Rashis S, Akhtar M. Uterine adenosarcoma with sarcomatous overgrowth and rhabdoid features: A rare case. Saudi J Med Med Sci. 2022; 10: 72-76.
- Dłuski D, Lewkowicz D, Leszczyńska-Gorzelak B, Obrzut B, Rechberger T, et al. An Unusual Coexistence of Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma with Endometrioid-Type Endometrial Cancer in a 58-Year-Old Woman: A Case Study with Literature Review. Case Rep Oncol. 2018; 31: 347-352.
- Vignali M, Messori P, Zacche MM, Natale A, Busacca M. Mullerian adenosarcoma: a frequently misinterpreted diagnosis. J Minim Invasive Gynecol. 2013; 20: 551.