Clinical & Medical Surgery
Open Access
Volume 3
Chukwuma Grant Madubuko; Chimaobi Tim Nnaji*; Ugochi Regina Onwudiwe
*Corresponding Author: Chimaobi Tim Nnaji
Consultant Anaesthetist, Department of Anaesthesia, Federal University Teaching Hospital Owerri-Imo State, Nigeria.
Tel: +2348064871001; Email: chymaoby@yahoo.com
Article Info
Received: Dec 26, 2022
Accepted: Jan 23, 2023
Published: Jan 31, 2023
Archived: www.jclinmedsurgery.com
Copyright: © Nnaji CT (2023).
Abstract...
Background: Epidural anaesthesia can be administered as single shot, intermittently, continuously or as patient controlled injection. Our study compared the intraoperative haemodynamic effects of continuous versus intermittent epidural injections of bupivacaine with fentanyl for major gynaecological surgeries.
Methods: A total of 50 female adult patients, age 30 - 70 years, ASA I, II or III were randomised into either Group C ( n=25), to received continuous epidural infusion of 10 ml mixture of 0.25% bupivacaine with fentanyl 2 μg/ml at the rate of 10 ml/hr or Group I (n=25), to received programmed intermittent epidural injection of 10 ml mixture of 0.25% bupivacaine with fentanyl 2 μg/ml given every 1 hour until the end of surgery. Both groups received an initial loading dose of 10 ml mixture of 0.5% bupivacaine (9 ml) with fentanyl 50 μg (1 ml). The primary outcome evaluated was the intraoperative haemodynamic parameters (HR, SBP, DBP and MAP). Data was collected and analysed with SPSS 21 for windows. A p-value of <0.05 was considered significant.
Results: The analysis of the intragroup haemodynamic parameters at different times showed statistically significant percentage decreases from the baseline; In Group I, the decrease in mean HR at 45th and 90th min was -2.9% (p>0.05) and -9.0% (p<0.01), while in Group C, -11.4% (p<0.01) and -14.4% (p<0.001) respectively. Changes in MAP from the baseline was also found to be more statistically significant in Group C ( -10.0%, p<0.001 and -13.8%, p<0.001) compared to Group I (-9.9%, p<0.01 and -14.1%, p<0.001) at the 45th and 90th min respectively. The mean total local anaesthetic consumption was significantly lower in Group I (18.4 ± 3.9 ml) compared with Group C (21.0 ± 2.6 ml), p=0.01. The incidence of Pruritus and Shivering observed during the study were comparable between the groups (p= 0.07 and p= 0.68 respectively).
Conclusion: Intermittent or continuous epidural bupivacaine-fentanyl injection provides stable and comparable haemodynamic effects, but with more variations in the patients receiving continuous epidural anaesthesia for major gynaecological surgeries.
Citation: Madubuko GC, Nnaji CT, Onwudiwe RU. Comparative Evaluation of Haemodynamic Effects of Continuous Versus Intermittent Epidural Injections of Bupivacaine with Fentanyl for Major Gynaecological Surgeries. J Clin Med Surgery. 2023; 3(1): 1071.
Introduction
Anaesthesia for gynaecological surgeries could be general, epidural, spinal or combined spinal-epidural anaesthesia [1]. Open abdominal myomectomy and hysterectomy are major types of gynaecological procedures and their anaesthesia poses some challenges, especially in developing countries due to socioeconomic factors like poverty, illiteracy, unavailability of anaesthetic drugs and insufficient number of trained Physician Anaesthetists [2,3]. Takai et al [2] found that a high number of the procedures are done under general anaesthesia, but they also noted the use of regional anaesthesia. In another study conducted by Nnaji et al [3], it was observed that combined spinal-epidural (CSE) anaesthesia offers some benefits in terms of better intraoperative and postoperative analgesia in abdominal Myomectomy, but the utilisation rate was very low (1.5%).
General anaesthesia offers better relaxation for gynaecological procedures; however, it could be associated with airway mishaps [4]. While spinal anaesthesia offers good analgesia in an awake patient, most time it does not provide anaesthesia long enough to last the duration of the surgery. Epidural anaesthesia, which is often under utilised in our environment, can offer both intraoperative and postoperative analgesia, and it has the potential to reduce or eliminate the perioperative physiologic stress response to surgery and thereby decrease surgical complications and improve outcomes [5]. Epidural anaesthesia is usually administered for surgeries in the lower abdomen, perineum and lower extremities. Although its onset of action is slow, and sometimes associated with patchy sensory blocks, when properly performed, it can offer good anaesthesia and outlast the duration of prolonged major gynaecological surgeries like abdominal myomectomy and hysterectomy [6,7].
Epidural anaesthesia can be administered as single shot, intermittently, continuously or as patient controlled injection. Some authors have evaluated the addition of adjuvants like dexmedetomidine, clonidine, morphine, fentanyl and neostigmine to local anaesthetic like bupivacaine for epidural anaesthesia in an attempt to potentiate and prolong the analgesic effect [8,9]. However, consideration should also be made to evaluate the haemodynamic effect of combination of local anaesthetics with adjuvants and the techniques of epidural anaesthesia.
There is a perception that continuous epidural infusion of local anaesthetic produces an unchanging block to maintain analgesia and minimise cardiovascular disturbance [10], but this has not been exclusively evaluated. Thus, we conducted a prospective study with the primary aim of determining the differences in the intraoperative haemodynamic changes using continuous or intermittent epidural injection of local anaesthetic agents with opioids. The secondary outcome measures were to determine the differences in the maximum level of sensory block, total volume of epidural drug injection, and side effects that can occur between the two methods of epidural administration.
Methodology
Ethical clearance for this prospective randomized single blind comparative study was obtained from the Health Research and Ethics Committee of our Institution. The study was conducted over a period of 6 months (June – November 2020). We recruited adult female patients, aged 30-70 years, ASA physical status I, II or III consented patients, booked for elective open abdominal myomectomy or hysterectomy. Those that refused to consent to the study, or with history of coagulopathy, deformity of the spine, allergy to either local anaesthetic agents or opioid, hypotension, cognitive impairment, sepsis were excluded from the study, as well as those that had inadequate epidural anaesthesia/analgesia, that were subsequently converted to general anaesthesia.
Preoperative evaluation was done at least a day before the surgery, to establish rapport with the patient, clinically ascertain fitness to undergo anaesthesia and surgery, establish pre-anaesthetic and postoperative plans, and obtain informed written consent.
Sample size was calculated using a significance level of 5%, power of study 90% and standard deviations for mean SBP in the 2 groups of Mogahed et al [11] and Rajarajeswaran et al [12] studies (continuous epidural injection of 0.5% bupivacaine SD = 11.3 mmHg and intermittent epidural injection of 0.5% bupivacaine SD = 7.53 mmHg respectively). About 22.24 patients were required in each group to demonstrate changes in SBP of significance. However, allowing for 10% attrition to make allowance for loss of follow-up/protocol violation, a total of 50 patients were used for the study.
On the morning of the surgery in the theatre, Patients were received on arrival, identified using the patient's tag and operation list, and transported to the gynaecological theatre on a trolley. The eligible patients were allocated into group C (n=25) and group I (n=25) in the surgical theatre reception area in a balanced manner with a pick from computer-generated number allotment that was concealed in a brown envelope, by a research assistant (Anaesthesia Registrar). In the operating room, the baseline pulse rate (PR), arterial oxygen saturation (SaO2), peripheral body temperature, non-invasive systolic, diastolic and mean arterial blood pressures (SBP, DBP, MAP) and electrocardiogram were checked and recorded using a multiparameter patient monitor (MEC-2000. Produced by Shenzhen Mindray BioMedical Electronics Co. LTD, China, April 2014). Intravenous access was gained with either a size 16 or 18-gauge cannula on the non-dominant hand, 4mg ondansetron was given for emesis prophylaxis and 10 ml/kg of warm 0.9% sodium chloride was infused over 20 minutes as preload for epidural injection. Intravenous maintenance fluid administration was given at the rate of 4 – 8 ml/kg/hour and/or guided by the haemodynamic status of the patient or blood loss. The WHO Surgical Safety Checklist was done before induction of epidural anaesthesia.
The epidural procedure, administration of study drugs and intraoperative patient management were done by the principal investigators, while the data collection was done by a trained research assistant (Anaesthesia Registrar) who was blinded to the groupings. Patient sits with their elbows resting on their thighs or hugs a pillow, arching the back with the help of an assistant to obtain a maximal spinal flexion. Under aseptic condition, the lower back of the patient was cleaned with povidone-iodine and draped. The Tuffier's line was used as landmark to identify the L3-L4 intervertebral space and through a midline approach, the skin and underlying structures were infiltrated with 3 ml of 1% lidocaine using 22- gauge hypodermic needle. Size 18- gaugeTuohy needle was introduced into the epidural space using the loss of resistance to air technique, and a multiport epidural catheter threaded cephalad through the needle to a depth of 6 cm into the epidural space. The Tuohy needle was then withdrawn. Following negative aspiration of cerebrospinal fluid (CSF) and blood, 3 ml of lidocaine with epinephrine 1:200,000 (test dose) was injected through the catheter, and evaluated for 5 minutes, to confirm correct catheter placement and exclusion of intravascular or intrathecal catheter placement. All the patients (Group C and I) received a loading dose of 10 ml mixture of 0.5% isobaric bupivacaine (9 ml), fentanyl 50 ug (1ml) titrated to effect in aliquot of 5 ml, to allow for individual differences. Then the epidural catheter was carefully strapped and secured on the patient's shoulder, and patient positioned supine for surgery with a pillow support below the head and the shoulders.
The SBP, DBP and MAP, HR, SaO2, electrocardiogram, temperature, level of sensory block, and degree of motor block, were monitored until the sensory and motor blocks reach dermatomal level T6 and modified Bromage's score 2 respectively. The level of sensory block was assessed using a piece of cotton wool soaked in ethyl alcohol (cold and heat sensation) to touch the patients extending to higher dermatomal levels every 2 minutes until the block reaches its maximum. The modified Bromage Scoring System (0= no weakness, 1= unable to raise extended leg, but move knees and feet, 2= unable to raise extended leg nor flex knee but able to move feet, 3= unable to move any joint in legs (complete block)) was used to assess the extent of motor block [13]. Having achieved the desired level of sensory block (dermatomal level = T6) and motor block (modified Bromage's Score = 2), intravenous midazolam 0.02 mg/kg was given for anxiolysis, and surgery commenced. Patients whom sensory level of T6 was not achieved even after 30 minutes of loading dose, or who had Visual Analogue Score (VAS) > 3 (1 = no pain, 10 = worst possible pain) following surgical stimulation and failed to respond to rescue dose of 5ml epidural injection of study agent were converted to general anaesthesia for the surgery, and excluded from the study. A VAS > 3 was considered significant for pain.
The onset of sensory block was defined as the time from the epidural injection to occurrence of sensory block level of T6 dermatome. The onset of motor blockade was defined as the time from epidural injection to occurrence of modified Bromage's score 2. On achieving the desired level of sensory block and motor block, continuous infusion of mixture of 0.25% bupivacaine with fentanyl 2 ug/ml at the rate of 10 ml/hr was commenced in Group C (n=25), while Group I received programmed intermittent epidural injection of 10 ml mixture of 0.25% bupivacaine with fentanyl 2 ug/ml every 1 hour till the end of surgery, with syringe pump (Graseby 3300, Smith Medical International Ltd, Hertfordshire, UK).
Intraoperatively, HR, SBP, DBP and MAP were monitored continuously and documented every 3 minutes in the first 30 minutes, thereafter every 15 minutes till the end of surgery, and a value not greater than 20% from the baseline was maintained. Hypotension [13] (for the purpose of this study was taken as SBP ≤ 90 mmHg or decrease of greater than 25% MAP from the baseline) and bradycardia [12] (heart rate less than 50 beats per minute) was treated with intravenous ephedrine 0.07 mg/kg and atropine 0.01 mg/kg respectively and documented. There was continuous monitoring of arterial oxygen saturation to ensure a value >95%, electrocardiography to ascertain a sinus rhythm, normal waves, intervals and segments, peripheraltemperature to maintain a normothermia (36.5–37.4oC), and intraoperative blood loss estimation. Desaturation was defined as a value <95% and was treated with 100% oxygen via nasal catheter. Other causes were excluded and normovolaemia and normothermia ensured. Hypothermia was prevented and managed by giving warm fluids and keeping the theatre temperature at 25˚C.
Subsequent monitoring of sensory block was done with cotton wool soaked in ethyl alcohol (cold and heat sensation) every 10 minutes until 2 segments regression from the highest level of sensory block, and this necessitated administration of top up epidural injection of 5 ml of study agent, especially if patient complains of pain (VAS > 3). Urine output was monitored to ensure a value of >0.5 ml/kg/hr. Sedation was assessed and recorded every 15 minutes using a six-point Ramsay sedation scale [13] (1= Anxious, agitated or restless, 2= Cooperative, oriented, and tranquil, 3= Responds to command, 4= Brisk response to light glabellar tap or loud auditory stimulus, 5= sluggish response, 6= No response). Side effects like shivering and pruritus were managed and documented. At the end of surgery, rectal diclofenac 100 mg was inserted, and the patients transferred to the post-anaesthetic care room (PACR). All the patients were adequately covered with blankets to prevent the occurrence of shivering which may result from heat loss.
On arrival to the PACR the SBP, DBP, MAP, SaO2, heart and respiratory rates, temperature, regression of sensory block and motor-block, pain and sedation were monitored and recorded at 0, 15 and 30 minutes, 1, 2, 3, and 4 hours and subsequently patient transferred to the gynaecology ward, where monitoring continued at the 8, 12, 16, 20 and 24th hour. The duration of sensory and motor blocks was determined from time of epidural injection of loading dose of study agent, to the regression of the sensory block to S1 dermatome and motor block regression to modified Bromage 0, and was recorded. Pain was assessed with the VAS. Patients with pain score >3 while in PACR and subsequently in the Gynaecology ward received top up of epidural analgesia of 5 ml of 0.125% bupivacaine every 4 hours until 24 hours period, after which the epidural catheter was removed. The duration of analgesia was taken as the interval between the loading dose of epidural injection of study agent to the time of first request for analgesia (VAS > 3) in PACR. The total volume of epidural injection given for each patient was documented.
The data collected was entered into a data collection form and analysed with the Statistical Package for Social Sciences (SPSS) 21 version for windows. Tables and figures were used to present the result, and expressed as mean, median (interquartile range), proportion (number of patients), and Standard Deviation (SD). Continuous data like age, weight, height, BMI, systolic and diastolic blood pressure, mean arterial pressure, heart rate, were compared using unpaired student's t test. Chi-squared test was used for test of significance between categorical variables such as incidence of side effects between the two groups. Paired student′s t test was used for intragroup analysis of the haemodynamic parameters. Mann–Whitney U test was used to compare the highest level of sensory block, the mean regression time of sensory block (S1) and mean regression time of motor block (M0). A p-value of <0.05 was considered significant.
Results
Fifty patients were recruited for this study; 25 patients in Group I and 25 patients in Group C, and all completed the study. Table I compares the demographic characteristics of the patients in groups I and C. The mean age was 41.80 ± 8.50 yr in Group I and 42.20 ± 10.90 yr in Group C, and this was not statistically significant (p=0.87). The mean weight, height and BMI in Group I (63.10 ± 7.30 kg, 1.62 ± 0.04 m and 23.8 ± 2.3 kg/m2) and Group C (63.80 ± 9.90 kg, 1.64 ± 0.08 m and 23.8 ± 3.0 kg/m2) were comparable with a p value of 0.77, p=0.34 and p=0.92 respectively.
The ASA I/II was 9/16 in Group I and 13/12 in Group C, (p=0.35). The mean duration of surgery was longer in Group C (132.30 ± 30.30 min) compared with Group I (127.40 ± 34.30 min), p=0.60. The mean total local anaesthetic consumption was significantly lower in Group I (18.4 ± 3.9 mg) compared with Group C (21.0 ± 2.6 mg), p=0.01 (Table 2).
Table 3 compared the sensory and motor block characteristics between Group I and Group C. The mean onset of sensory and motor blocks was faster in Group I (19.30 ± 5.70 min and 11.30 ± 3.40 min), compared to Group C (20.80 ± 5.00 min and 12.90 ± 3.80 min), but the differences were respectively comparable (p=0.32 and p=0.12). The median maximum sensory block height was T4 (T4 – T6) in Group I and T4 (T4 – T6) in Group C, p=0.05. The mean duration of sensory block comparable (p=0.29) between Group C (320.84 ± 84.69 min) and Group I (305.68 ± 86.33 min). The mean duration of motor block (regression of motor block to modified Bromage 0) was longer in Group C (231.16 ± 48.06 min), compared to Group I (228.36 ± 55.94 min), but the difference was not statistically significant (p=0.63).
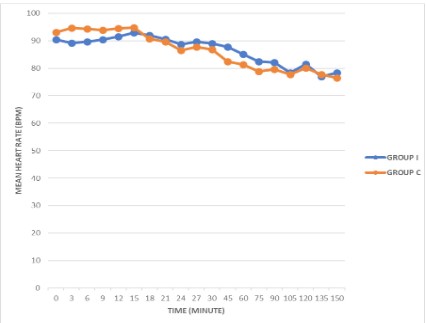
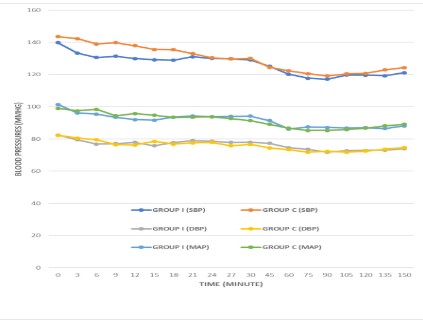
The comparative distributions of the mean HR, SBP, DBP and MAP of the groups are highlighted in Figures 1 and 2, while Table IV and V Shows the percentage intragroup changes of the mean HR, SBP, DBP, MAP from the baseline in Group I and Group C respectively.
The incidence of pruritus and shivering was observed to be respectively 12% and 12% in Group C compared to 0% and 16% in Group I, p=0.07 and p=0.68 respectively (Table 6).
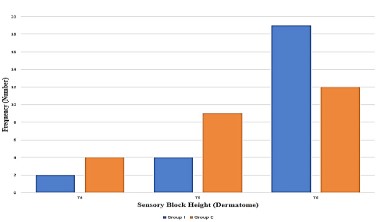
Figure 3 shows the distribution of maximum sensory block height between Group I and Group C. The maximum sensory block height of T4 was observed more in patients in Group C (4) compared to Group I (2). The sensory block height of T5 was also more in Group C (9) compared to Group I (4). However, the maximum sensory block height of T6 was observed in 19 patients in Group I compared to 12 patients in Group C.
Table 1: The demographic characteristics of patients in the Intermittent Group and Continuous Group.
| Variables | Group I (n=25) Mean ± SD | Group C (n=25) Mean ± SD | p value |
|---|---|---|---|
| Age (yr) | 41.8 ± 8.5 | 42.2 ± 10.9 | 0.87 |
| Weight (kg) | 63.1 ± 7.3 | 63.8 ± 9.9 | 0.77 |
| Height (m) | 1.62 ± 0.04 | 1.64 ± 0.08 | 0.34 |
| BMI (kg/m2) | 23.8 ± 2.3 | 23.8 ± 3.0 | 0.92 |
Group I: Intermittent Group; Group C: Continuous Group; SD: standard deviation.
Table 2: The ASA and intraoperative variables of the patients in the Intermittent Group and Continuous Group.
| Variables | Group I (n=25) Mean ± SD | Group C (n=25) Mean ± SD | p value |
|---|---|---|---|
| ASA Status(I/II/III) | 9(36.0)/16(64.0)/0(0) | 13 (52.0)/12 (48.0)/0 (0) | 0.35 |
| Duration of surgery(min) | 127.4±34.3 | 132.3 ± 30.3 | 0.60 |
| Total local anaesthetic volume (ml) | 18.4 ± 3.9 | 21.0 ± 2.6 | 0.01* |
*Indicates significant difference between groups p< 0.05; Group I: Intermittent Group; Group C: Continuous Group; SD: standard deviation.
Table 3: The sensory and motor block characteristics observed in the Intermittent Group and the Continuous Group.
| Variables | Group I (n=25) | Group C (n=25) Mean ± SD | p value |
|---|---|---|---|
| Onset time of sensory block (min) | 19.30±5.70 | 20.80±5.00 | 0.32 |
| Onset time of motor block (min) | 11.30±3.40 | 12.90±3.80 | 0.12 |
| Maximum sensory block height | T4(T4–T6) | T4(T4–T6) | 0.05 |
| Duration of sensory block(min) | 305.68±86.33 | 320.84±84.69 | 0.29 |
| Duration of motor block(min) | 228.36±55.94 | 231.16 ± 48.06 | 0.63 |
| Duration of analgesia (min) | 259.80±83.30 | 327.90±127.90 | 0.03* |
*Indicates significant difference between groups p< 0.05; Group I: Intermittent Group; Group C: Continuous; Group; SD: standard deviation.
Table 4:Percentage intragroup changes of the mean HR, SBP, DBP, MAP from the baseline in Group I.
| Group C N=25 | Baseline | 6 min | 12 min | 18 min | 24 min | 30 min | 45 min | 60 min | 75 min | 90 min | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| HR(b/min) | 93.1±17.9 | 94.4±18.1+1.5% | 94.5±19.0+1.5% | 90.8±19.9-2.5% | 86.5±15.3-7.1%¶ | 86.8±15.5-6.8%¶ | 82.5±15.3-11.4%§ | 81.4±14.3-12.5%§ | 78.8±13.6-15.3%† | 79.6±14.4-14.4%† | |
| SBP (mmHg) | 143.6±19.2 | 139.0±18.6-3.2%¶ | 138.0 ±20.2-3.9%¶ | 135.5 ± 18.4-5.6%§ | 130.5 ± 18.4-9.1%§ | 130.1 ± 21.4-9.4%§ | 124.5 ± 19.6-13.3%† | 122.4 ± 23.1-14.8%† | 120.6 ± 20.1-16.0%† | 119.2 ± 18.8-17.0%† | |
| DBP(mmHg) | 82.3±10.1 | 79.6±9.3-3.2% | 76.3 ± 12.5-7.3%¶ | 76.8 ± 11.6-6.7%¶ | 77.9 ± 13.4-5.3% | 76.7 ± 15.2-6.8% | 124.5 ± 19.6-13.3%† | 74.4 ± 13.4-9.5%¶ | 73.4 ± 14.8-10.8%¶ | 71.8 ± 13.7-12.8%§ | 72.3 ± 11.7-12.2%§ |
| MAP(mmHg) | 99.0±11.7 | 98.4±13.3-0.6% | 95.8±14.4-3.2% | 93.5±13.5-5.5% | 93.7±13.4-5.4% | 91.4 ± 17.4-7.7% | 89.1 ± 6.8-10.0%† | 86.7 ± 17.6-12.4%† | 85.3 ± 16.2-13.8%† | 85.3 ± 14.3-13.8%† |
¶p<0.05, §p<0.01, †p<0.001. HR, Heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure.
Table 5:Percentage intragroup changes of the mean HR, SBP, DBP, MAP from the baseline in the Continuous Group.
| Group C (N=25) | Baseline | 6 min | 12 min | 18 min | 24 min | 30 min | 45 min | 60 min | 75 min | 90 min | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| HR(b/min) | 93.1±17.9 | 94.4±18.1+1.5% | 94.5±19.0+1.5% | 90.8±19.9-2.5% | 86.5±15.3-7.1%¶ | 86.8±15.5-6.8%¶ | 82.5±15.3-11.4%§ | 81.4±14.3-12.5%§ | 78.8±13.6-15.3%† | 79.6±14.4-14.4%† | |
| SBP (mmHg) | 143.6±19.2 | 139.0±18.6-3.2%¶ | 138.0 ±20.2-3.9%¶ | 135.5 ± 18.4-5.6%§ | 130.5 ± 18.4-9.1%§ | 130.1 ± 21.4-9.4%§ | 124.5 ± 19.6-13.3%† | 122.4 ± 23.1-14.8%† | 120.6 ± 20.1-16.0%† | 119.2 ± 18.8-17.0%† | |
| DBP(mmHg) | 82.3±10.1 | 79.6±9.3-3.2% | 76.3 ± 12.5-7.3%¶ | 76.8 ± 11.6-6.7%¶ | 77.9 ± 13.4-5.3% | 76.7 ± 15.2-6.8% | 124.5 ± 19.6-13.3%† | 74.4 ± 13.4-9.5%¶ | 73.4 ± 14.8-10.8%¶ | 71.8 ± 13.7-12.8%§ | 72.3 ± 11.7-12.2%§ |
| MAP(mmHg) | 99.0±11.7 | 98.4±13.3-0.6% | 95.8±14.4-3.2% | 93.5±13.5-5.5% | 93.7±13.4-5.4% | 91.4 ± 17.4-7.7% | 89.1 ± 6.8-10.0%† | 86.7 ± 17.6-12.4%† | 85.3 ± 16.2-13.8%† | 85.3 ± 14.3-13.8%† |
¶p<0.05, §p<0.01, †p<0.001. HR, Heart rate; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure.
Table 6:The Incidence of Side Effects observed in the Intermittent Group and the Continuous Group.
| Intermittent Group | Continuous Group | ||||
|---|---|---|---|---|---|
| Side Effects | Yes n(%) | No n(%) | Yes n(%) | No n(%) | p value |
| Shivering | 4(16.0) | 21(84.0) | 3(12.0) | 22(88.0) | 0.68 |
| Pruritus | 0(0.0) | 25(100.0) | 3(12.0) | 22(88.0) | 0.07 |
Discussion
This study has shown that the administration of epidural bupivacaine-fentanyl either through intermittent or continuous method, provides stable haemodynamic effects irrespective of the sensory block height for major gynaecological surgeries.
Epidural injection of 0.25% bupivacaine with fentanyl 2ug/ml either intermittently or through a continuous method provided comparable and stable intraoperative heart rates in this study. This is similar to the findings of Duncan et al [14], who reported that continuous and intermittent epidural injection of bupivacaine provided stable heart rate. In addition, Shidhaye et al [15] in their study of women receiving epidural labour analgesia reported that the administration of both CEA and IEA provided stable and comparable heart rate in their subjects. Heart rate is influenced by the autonomic nervous system and a lower heart rate indicates parasympathetic predominance [16].
The heart rate variability associated with epidural anaesthesia is complex and it can be related to the level of anaesthesia, amount of local anaesthetic injected, the addition of epinephrine to the local anaesthetic solution and the clinical condition of the patient. The stability in heart rate observed in our study could be related to the type of patients recruited for the study. We evaluated ASA I, II and medically stable ASA III patients. The stability in heart rate we observed corroborates with the find ing of Twardowski et al [17], that epidural anaesthesia and the sympathetic blockade associated with the technique do not significantly affect the electrical functions of the cardiac atria reflected in superficial ECG. Epidural anaesthesia has been reported to significantly affects the coronary flow via vasodilatation and brings about the improvement of the coronary flow during diastole [17].
Bradycardia following epidural injection of local anaesthetic results from the blockade of sympathetic cardiac accelerator fibres and decreased venous return to the heart [18]. Bradycardia for the purpose of this study was described as heart rate lower than 50 beats per minute was not observed in this research. This could be attributed to the proper patient's selection and preparation as no patient in both groups had low baseline heart rates. It has been reported that epidural block at the level of mid thoracic and lumbar region (T5-L4) results in peripheral sympathetic and splanchnic fibres blockade. Following this, there is vasodilatation in the blocked areas with compensatory vasoconstriction of the capacitance vessels in the remaining unblocked areas [19]. However, the degree of compensatory vasoconstriction is said to be dependent on the upper level of analgesia which is mediated mainly by means of baroreceptors via those sympathetic vasoconstrictor nerves (T1-T5) that remain unblocked [20]. This could also explain the stability in heart rate seen in both groups in our study as the highest level of epidural blockade observed in both groups was T4, but an intragroup analysis of the changes in the heart rates from the baseline during the study period showed some degree of significance at different times as shown in Table IV and V.
In patients that received programmed intermittent epidural anaesthesia, there were percentage decreases in mean HR noted from the baseline after induction of epidural anaesthesia, but these decreases were not statistically significant until the 75th and 90th minute. This later significant decrease occurred possibly as a result of the 1st dose of the study agent given at the 60th minute. This is similar to the findings in Hammond et al [20] study who reported the haemodynamic effects of induction of epidural analgesia in women in labour using 7 to 10 ml of 0.5% plain bupivacaine. At 30 and 45 minutes, they observed a decrease of -4.7% and -2.4% respectively in the mean HR when compared with the baseline value which was not statistically significant. However, their study period did not exceed 45 minutes. In the continuous epidural group, after epidural induction and commencement of continuous infusion, it was noted that there was a significant decrease in the mean HR from the baseline at the 24th, 45th, 60th, 75th and 90th minute. This level of significant decrease seen could be related to the proportion of high-level epidural blockade seen in patients that received continuous epidural anaesthesia, where 4 of the patients had T4 and 9 patients T5 sensory blockade, while in patients that received intermittent programmed epidural anaesthesia, 2 patients had T4, and 4 patients T5 sensory blockade. This means that those that received intermittent programmed epidural anaesthesia had more patients (19) with unblocked sympathetic vasoconstrictor nerves (T1-T5). But on the contrary, Duncan et al [14] reported that the epidural block in their study was more extensive in the intermittent group when compared to the continuous infusion group, however they found no differences in the cardiovascular parameters between the groups.
In this study, both groups were observed to have stable and comparable SBP, DBP and MAP, irrespective of the administration of CEA or IEA. This is consistent with the finding of Mogahed et al [11] and Prakash et al [21]. They noted that CEA or IEA provided stable and comparable blood pressures. Hammond et al [20] and Veering et al [19] reported that cardiovascular effects of epidural anaesthesia are proportional to the volume of local anaesthetic and height of block, and these effects result from denervation of the sympathetic outflow tracts. This produces dilatation of resistance and capacitance vessels and results in changes in the SBP, DBP and MAP of the patients.
It has been reported that the extent of denervation, balance of sympathetic and parasympathetic activity can influence patient's SBP, DBP and MAP. Also, individual response to different level of sympathetic blockade varies widely, depending on the degree of sympathetic tone before the epidural block. Thus, epidural anaesthesia limited to T5 and below, will result in vasodilatation of the pelvis and lower limb vessels. However, epidural block beyond T5 results in blocks of cardiac afferent and efferent fibres, leading to loss of chronotropic and inotropic drive to the myocardium. This can lead to blood pressure variability [17,18]. Virmani et al [23] found no significant fall in blood pressure in their study that compared continuous infusion of 0.166% bupivacaine at the rate of 5 ml/hr and 3 hourly bolus doses of 15 ml of same dose of bupivacaine for postoperative analgesia following renal surgery. They reported that this may be due to the low concentration of bupivacaine and continuous infusion of intravenous fluid given to the patients throughout the study period. In our study, 0.25% bupivacaine with fentanyl at the rate of 10 ml/hr (for both intermittent and continuous group) was used for intraoperative anaesthesia management of abdominal hysterectomy and myomectomy with the same comparable results in both groups. This may also be attributed to the concentration of bupivacaine used in our study.
Hypotension, in our study was described as systolic blood pressure (SBP) ≤90 mmHg or decrease of greater than 25% MAP from the baseline. Epidural injection of bupivacaine-fentanyl can result in cephalad spread of the drugs. This produces neuronal transmission blockade that results in decrease in sympathetic tone of blood vessels. The decreased vasomotor tone leads to the dilatation of the arterioles and pooling of blood in the capacitance vessels, thus, resulting in hypotension [20]. Campagna et al [24] reported that the main cause of hypotension following epidural anaesthesia is the decrease in the sympathetic outflow causing arterial vasodilatation, a decrease in venous return and consequently resulting in the activation of the Bezold Jarish reflex. Bezold Jarish reflex elicits a triad of bradycardia, vasodilatation and further hypotension by activation of 5-HT3 receptors within the intracardiac vagal nerve endings.
A further analysis of the haemodynamic parameters done within the groups in our study showed some significant percentage decrease from the baseline at different time interval, however, this was not enough to reflect hypotension in the mean difference. This is similar to the findings of Hammond and his colleagues [20], however, in their study they noted significant decrease in the mean SBP only at the 30th and 45th min. The percentage decreases in the mean DBP recorded at the 60th, 75th, 90th min, and the mean MAP recorded at the 12th, 18th, 24th, 30th, 45th, 60th, 75th, 90th min in intermittent epidural anaesthesia group were significantly low when compared with the baseline value. But in continuous epidural anaesthesia group, the significant difference in the intragroup mean DBP were observed in the measurements recorded at the 12th, 18th, 45th, 60th, 75th and 90th min, while that of mean MAP was noted at the 45th, 60th, 75th and 90th min. This variability in the in the intragroup SBP, DBP and MAP could be associated with periods of maximal epidural block height. In a study conducted by Baron et al [19], they noted that 30 minutes after the second epidural injection of bupivacaine, the upper level of anaesthesia was significantly higher and ranged from T9 to T6, and this also reflected as significant decrease in SBP, DBP and MAP measured at those time. According to them, the observed decrease in SBP, DBP and MAP occurred following a decrease in the right atrial pressure.
Hypotension due to epidural injection of local anaesthetic can be treated with intravenous fluid and or ephedrine [25]. Some measures taken to prevent hypotension in this study include preloading with intravenous fluid prior to the induction of epidural anaesthesia, as well as maintenance fluid therapy. Blood loss was also managed with fluid therapy where indicated.
The time taken to reach T6 sensory block were similar between the patients that received epidural anaesthesia via IEA and CEA technique. Although, the maximum height of block achieved was T4 in both groups, most of the patients' epidural block was limited to T5 and T6. Thus, this can explain why there were no significant variation in the SBP, DBP and MAP. In addition, none of the patients that participated in this study were found to have severe cardiovascular disease. The total volume of epidural bupivacaine-fentanyl injection was significantly more in the CEA patients compared to the IEA patients. This could be responsible for more extensive block seen in CEA group with more significant percentage decreases seen in the HR and DBP when compared with the baseline. Verghese et al [26] reported that the efficacy of epidural block is related to the concentration of local anaesthetic and its volume. Kleinman et al [27] reported that epidural injection for safety purposes, should be placed at an appropriate level that corresponds to the dermatome level of the intended surgical procedure. In the present study, epidural injection was performed at L3/L4 intervertebral space, and block height of T4 -6 were used for the surgeries.
The mean total volume of epidural injection used in this study was 18.4 ml in patients that received IEA and 21 ml in patients that received CEA. This volume of epidural bupivacaine (0.25%) was combined with fentanyl (2 μg/ml), and it provided adequate anaesthesia/analgesia throughout the period of study. It has been shown that the combination of neuraxial local anaesthetics with opioids provides anaesthesia [28]. Opioids work in the intrathecal space by activating opioid receptors in the dorsal grey matter of the spinal cord which modulates the function of afferent fibres. Fentanyl is commonly co-administered with bupivacaine for epidural analgesia/anaesthesia. Combining bupivacaine-fentanyl has the advantage of reducing the dose/volume that would be necessary if either drug were used alone, thus potentially decreasing the incidence and severity of associated side effects such as hypotension and motor block [29].
The patients that received CEA had longer duration of analgesia, sensory and motor blocks, compared with the patients that received IEA. Also, the volume of epidural injection was more with CEA. This agrees with the report of Higuchi et al [30], that the duration of sensory and motor blocks in a patient after epidural anaesthesia is a factor of the degree of the sensory and motor blocks, and this could be related to the volume of local anaesthetic causing prolonged sensory and intense motor blocks.
Though, this study was not structured to evaluate pain, there was no incidence of pain throughout the period of the study. Effective pain control during surgery is an essential component of patient care. When Chew et al [31] evaluated patients' knowledge of anaesthesia and perioperative care, they reported that pain was a common complaint of the patients they evaluated. The quality of intraoperative analgesia provided by CEA and IEA in this study was adequate for surgical anaesthesia throughout the study period, therefore, the need for top up doses of the study agent was insignificant in both groups looking at the total volume (LA) used and duration of surgery.
Epidural anaesthesia is frequently associated with shivering. The patients that received IEA had an incidence rate of 16%, while those that received CEA had a lower incidence rate of 12%, however, there was no statistical significance when both groups were compared (p<0.684). This is similar to the findings of Prakash et al [21]. In their study they evaluated the effects of 0.25% bupivacaine alone and with fentanyl or dexmedetomidine. They found the incidence of shivering of 12% in the bupivacaine-fentanyl group, 12% in the bupivacaine alone group and 4% in the bupivacaine-dexmedetomidine group, though no reason was given for this result. Shivering during surgery can cause some discomfort, interfere with patient's monitoring and at times exacerbates surgical pain. The mechanism of shivering under epidural anaesthesia is not clear. Hynson et al32 reported that shivering may be a normal thermoregulatory mechanism in response to a drop, in temperature due to redistribution of heat from core to the periphery. Shivering following epidural anaesthesia can occur as a result of fall in body temperature of about 1-3oC, which is probably due to the loss of thermo-sensory inputs, and heat loss from vasodilated anaesthetised areas.
In our study, there was no incidence of hypothermia. This could be related to the operating room temperature which was maintained at 24°C to 25°C, and the use of warmed intravenous crystalloid solutions and irrigation fluids during the procedures. All patients were premedicated with intravenous ondansetron, and those who had shivering were given oxygen therapy and intravenous paracetamol. Safavi et al [33] reported that intravenous ondansetron can decrease intensity and incidence of shivering without any haemodynamic side effect. In our study data on temperature was not documented, therefore correlation between shivering and temperature couldn't be established.
Another adverse effect observed in this study is pruritus. It is a known adverse effect of neuraxial narcotics. The exact mechanism is yet to be determined. The relationship of fentanyl with activation of supraspinal and dorsal horn mu receptors has been suggested [34]. An incidence as high as 95% has been reported following neuraxial fentanyl injection [35]. Antihistamines have been used in an attempt to reduce the incidence of neuraxial opiate-induced pruritus with varying degrees of success [36]. The incidence of 12% was reported only in CEA patients in this study. The effect was mild and well tolerated, requiring no treatment. Other complications like nausea, vomiting and desaturation were not observed. Our limitation is that we did not assay the plasma level of the medication used (bupivacaine + fentanyl), nor measure the cardiac output, cardiac index and stroke volume, because of dearth of the required equipment.
Conclusion
This study shows that when epidural bupivacaine-fentanyl is administered either through intermittent or continuous method, it provides stable and comparable haemodynamic effects, sensory block height and comparable incidence of side effects for major gynaecological surgeries. We recommend that continuous and intermittent epidural anaesthesia with bupivacaine-fentanyl should be used for major gynaecological surgeries, with a preference for intermittent method as it provides more stable haemodynamic effects with lesser volume of local anaesthetic agent with fentanyl.
References
- Bajwa SJ, Kulshrestha A. Anaesthesia for laparoscopic surgery: general vs regional anaesthesia. J Minim Access Surg. 2016; 12: 4-9.
- Takai IU, Yakasai IA, Omeje IJ, Ugwa EA. An audit of gynaecological procedures performed at Aminu Kano Teaching Hospital, Kano. J. basic clin. reprod. Sci. 2015; 4: 64-68.
- Saddler J. Gynaecological surgery. In: Allma KG, Wilson IH, editors. Oxford Handbook of Anaesthesia. 3rd ed. Oxford. oxford University Press. 2011; 613-629.
- Bauchat JR, Habib AS. Evidence-Based Anaesthesia for major gynaecologic surgery. Anesthesiol Clin. 2015; 33: 173-207.
- Nnaji CT, Chikwe K. Anaesthesia for abdominal myomectomy – A five years audit of a federal medical center in Owerri, Nigeria. J Anest Anesthes. 2017; 1: 16-19.
- Cook TM, MacDougall–Davis SR. Complications and failure of airway management. Br J Anaesth. 2012; 109: 68-85.
- Park WY, Thompson JS, Lee KK. Effect of epidural anesthesia and analgesia on perioperative outcome. Ann. Surg. 2001; 234: 560- 571.
- Yin XK, Xiang ZG, Prasia A, Lamichhane N. Continuous versus intermittent bolus epidural anaesthesia in elderly surgical patients. J Nepal Med Assoc 2003; 42: 201-203.
- Gupta P, Dua CK, Verma UC, Saxena KN, Chakraborty I. Sequential combined spinal epidural versus epidural anaesthesia in orthopaedic and gynaecological surgery: a comparative evaluation. Indian J Anaesth. 2002; 46: 453-456.
- Silva M, Halpern SH. Epidural analgesia for labour: current techniques. Local Reg Anesth. 2010; 3: 143-153.
- Mogahed MM, Hamed JE, Mohammad Refaat HS. Epidural Anesthesia versus General Anesthesia in Uterine Artery Embolization. J Anesth Clin Res. 2017; 8: 761.
- Rajarajeswaran K, Sivakumar S. A comparative study of epidural fentanyl and clonidine as an adjunct to bupivacaine in patients undergoing abdominal hysterectomy. Int J Med Health Res. 2016; 2: 35-39.
- Swain A, Nag DS, Saliu S, Sammader DP. Adjuvants to local anesthetics. Current understanding and future trends. World J Clin Cases. 2017; 5: 307 - 323.
- Duncan LA, Fried MJ, Lee A, Wildsmith JAW. Comparison of continuous and intermitted administration of extradural bupivacaine for analgesia after lower abdominal surgery. Br J Anaesth. 1998; 80: 7 -10.
- Shidhaye RV, Badhe V, Divekar D, Dhulkhed V, et al. A randomized clinical trial to compare small frequent boluses technique with that of traditional intermittent top-ups and continuous epidural infusion, for maintenance of epidural labour analgesia. Sri Lankan J. Anaesthesiol. 2010; 26: 75-80.
- O’Neill J, Bounford K, Anstey A, D’Silva J, Clark L, Plein S, et al. P wave indices, heart rate variability and anthropometry in a healthy South Asian population. Plos one. 2019; 14: e0220662.
- Twardowski P, Owczuk R, Wujtewicz MA, Wojciechowski J, et al. The influence of epidural anesthesia on the electrical activity of heart atria. Kardiochir. Torakochirurgia Pol. 2014; 11: 156-161.
- Veering BT, Cousins MJ. Cardiovascular and pulmonary effects of epidural anaesthesia. Anaesth Intensive Care. 2000; 28: 630- 635.
- Baron J-F, Payon D, Corlat P, Edouard A, Viars P. Forearm vascular tone and reactivity during lumbar epidural anesthesia. Anesth Analg. 1988; 67: 1065-1070.
- Hammond RR, Webster AC. Haemodynamic effects of induction of epidural analgesia in labour. Can J Anaesth. 1988; 35: 41-46.
- Prakash R, Kushwaha BB, Shashibhushan, Bhatia VK, Chandra G, Singh BP. A comparative study of bupivacaine 0.25% alone and with fentanyl or dexmedetomidine for percutaneous neph- rolithotomy (PCNL) under epidural anaesthesia. Indian J Sci Res 2014; 5: 39-46.
- Veering BT, Cousins MJ. Cardiovascular and pulmonary effects of epidural anaesthesia. Anaesth Intensive Care. 2000; 28: 630- 635.
- Virmani R, Ghai A, Singh DK. A study to compare continuous epidural infusion and intermittent bolus of bupivacaine for postoperative analgesia following renal surgery. South. Afr J Anaesth Analg. 2008; 14: 19-22.
- Campagna JA, Carter C. Clinical relevance of the Bezold-Jarisch reflex. Anesthesiology. 2003; 98:1250-1260.
- Holte K, Foss NB, Svensén C, Lund C, Madsen JL, Kehlet H. Epidural anesthesia, hypotension, and changes in intravascular volume. Anesthesiology. 2004; 100: 281-286.
- Verghese ST, Raafat SH, Jo Rice L, Belman AB, Patel KM. Caudal anaesthesia in children: effect of volume versus concentration of bupivacaine on blocking spermatic cord traction response during orchidopexy. Anesth Analg. 2002; 95: 1219-1223.
- Kleinman W, Mikhail M. Spinal, epidural, & caudal blocks. In: Morgan GE, Mikhail MS, Murray MJ, editors. Clinical Anesthesiology. 4th ed. New York: Lange Medical Books/ McGraw-Hill medical publishing division. 2006; 289-323.
- Ngan Kee WD, Khaw KS, Karman KL, So R. Synergistic Interaction between Fentanyl and Bupivacaine Given Intrathecally for Labor Analgesia. Anesthesiology. 2014; 120: 1126-1136.
- Comparative Obstetric Mobile Epidural Trial (COMET) Study Group UK. Effect of low-dose mobile versus traditional epidural techniques on mode of delivery: a randomised control trial. Lancet 2001; 358: 19-23.
- Higuchi H, Adachi Y, Tomiei Kazama T. Factors Affecting the Spread and Duration of Epidural Anesthesia with Ropivacaine. Anesthesiology. 2004; 101: 451-460.
- Chew ST, Tan T, Tan SS, IP-yam PC. A survey of Patients’ Knowledge of Anaesthesia and Perioperative Care. Singapore Med. J. 1998; 39: 399-402.
- Hynson JM, Sessler DI, Glosten B, McGuire J. Thermal balance and tremor patterns during epidural anesthesia. Anesthesiology. 1991; 74: 680-690.
- Safavi M, Honarmand A, Negahban M, Attari M. Prophylactic effects of intrathecal Meperidine and intravenous Ondansetron on shivering in patients undergoing lower extremity orthopedic surgery under spinal anesthesia. J Res Pharm Pract. 2014; 3: 94-99.
- Ackerman WE, Juneja MM, Kaczorowski DM, Colclough GW. A comparison of the incidence of pruritus following epidural opwww.jclinmedsurgery.com 9 oid administration in the parturient. Can J Anaesth. 1989; 36: 388-391.
- Wells J, Paech MJ, Evans SF. Intrathecal fentanyl-induced pruritus during labour: the effect of prophylactic ondansetron. Int J Obstet Anesth. 2004; 13: 35-39.
- Kumar K, Singh SI. Neuraxial opioid-induced pruritus: An update. J Anaesthesiol Clin Pharmacol. 2013; 29: 303-307.